Quick Summary: Yes, water can be made by combining hydrogen and oxygen atoms through combustion, creating H2O. However, the process is highly dangerous, energy-intensive, and impractical for large-scale production. Existing water reuse technologies and conservation methods are far safer and more cost-effective solutions to water scarcity than attempting to manufacture water from scratch.
The question seems simple enough. Water is just H2O—two hydrogen atoms bonded to one oxygen atom. We know the formula. We’ve mastered far more complex chemistry. So why can’t scientists just make water to solve global water shortages?
The answer is more complicated than the chemistry might suggest. And more explosive.
The Basic Chemistry of Water Creation
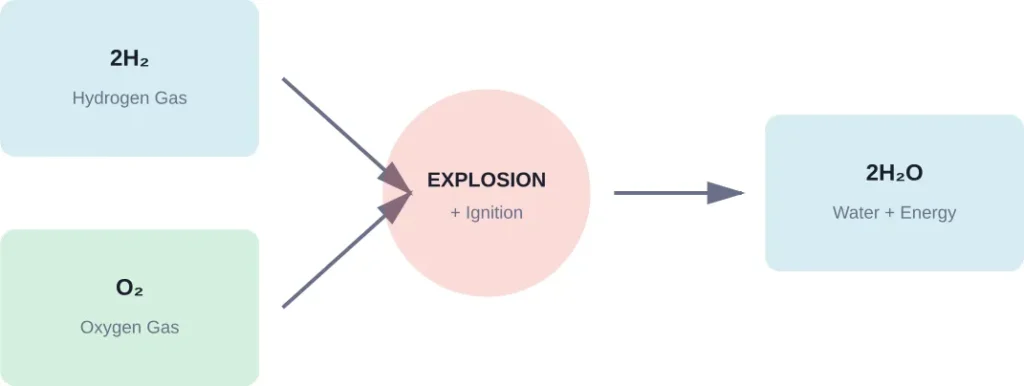
Water molecules form when hydrogen and oxygen combine through a chemical reaction. The process looks straightforward on paper: 2H₂ + O₂ → 2H₂O.
When hydrogen gas meets oxygen gas in the presence of an ignition source, they react violently to create water. The reaction releases tremendous energy in the form of heat and light. That’s exactly what happens during combustion.
But here’s the thing—this isn’t a gentle laboratory procedure. The reaction between hydrogen and oxygen is extremely dangerous and highly explosive.
Why Making Water Is Dangerously Impractical
The Hindenburg disaster of 1937 demonstrated exactly what happens when hydrogen combusts in the presence of oxygen. The airship burned catastrophically, killing 36 people in seconds.
That’s the same reaction scientists would need to harness to create water artificially. Not exactly a safe or scalable solution.
The Energy Problem
Creating water from elemental hydrogen and oxygen requires handling two highly volatile gases. Hydrogen is the most flammable element. Oxygen accelerates combustion.
The energy released during the reaction is substantial. While that energy could theoretically be captured, the infrastructure required would be massive. And dangerous.
There’s another issue. Where do the hydrogen and oxygen come from? They don’t exist freely in nature in the quantities needed.
Breaking Down Water to Make Water
Here’s the ironic part: the most accessible source of hydrogen and oxygen is water itself. Through electrolysis, electric current splits water molecules into hydrogen and oxygen gases.
But this process requires significant energy input. Then, to recombine those gases into water, the explosive reaction occurs. The net result? Energy wasted, danger introduced, and the same amount of water at the end.
It’s a closed loop that makes no practical sense.
What Science Is Actually Doing About Water Scarcity
Instead of manufacturing water from scratch, researchers and engineers focus on practical solutions that work today.
Industrial Water Reuse
According to EPA data, industrial water use is estimated to be more than 18.2 billion gallons per day from direct water withdrawals, not including water from public water supply. That represents enormous potential for recycling and reuse.
Industries are increasingly adopting water reuse technologies. Treated municipal wastewater serves in manufacturing processes, cooling systems for data centers, and other industrial applications. The same water circulates multiple times rather than being discarded after single use.
Advanced Filtration and Desalination
Water covers about 71% of Earth’s surface, with seas and oceans making up about 96.5% of water volume. The challenge isn’t creating new water—it’s making existing water usable.
Desalination technologies remove salt from seawater. Recent developments include artificial water channels that mimic Aquaporin proteins, potentially transporting water at lower energy costs than traditional methods.
These artificial channels have osmotic water permeability in the range of natural transporters, offering improved stability and simpler fabrication compared to biological proteins.

Water Harvesting Technologies
Atmospheric water harvesting extracts moisture from air. Floatable artificial photocatalysis devices can purify water while processing CO₂ from the environment simultaneously.
These technologies work with water that already exists rather than attempting dangerous chemical synthesis.
The Real Solution to Water Scarcity
The Earth’s population continues growing. Demand for water increases accordingly. But the answer isn’t making more water—it’s using what exists more wisely.
| Approach | Feasibility | Safety | Cost | Scalability |
|---|---|---|---|---|
| Manufacturing water from H₂ + O₂ | Low | Dangerous | Extremely High | Impractical |
| Industrial water reuse | High | Safe | Moderate | Excellent |
| Desalination | High | Safe | High | Good |
| Water conservation | Very High | Safe | Low | Excellent |
Conservation remains the most cost-effective strategy. Reducing waste in industrial facilities, fixing infrastructure leaks, and implementing efficient irrigation saves billions of gallons.
According to EPA data on water use at industrial facilities, identifying and eliminating water waste represents massive opportunity. Water is necessary for all types of industry, making efficiency improvements immediately valuable.
Frequently Asked Questions
Yes, water forms when hydrogen and oxygen gases react. However, the reaction is explosive and requires careful control. Labs demonstrate this in small-scale experiments, but it’s not practical for producing drinking water.
Making water from hydrogen and oxygen is far more expensive and dangerous than purifying, recycling, or transporting existing water. The energy costs alone make it impractical compared to desalination or conservation.
Most hydrogen and oxygen for industrial use comes from splitting water molecules through electrolysis. This means using water to make the components to make water—a pointless cycle that wastes energy.
No. Water molecules created through hydrogen-oxygen combustion are chemically identical to natural water. Both are H2O. The difference is the process, not the product.
Artificial diamonds are made from carbon under extreme pressure. The process is expensive but doesn’t involve explosive reactions. Carbon is also abundant and stable. Water creation requires handling two of the most reactive gases known, making the comparison imperfect.
In space exploration, hydrogen-oxygen fuel cells create water as a byproduct of power generation. Astronauts benefit from both electricity and drinking water. But this works because spacecraft already carry these fuels for propulsion.
Recycling wastewater, improving infrastructure to prevent leaks, implementing efficient irrigation, and advancing desalination technology. These methods work with existing water rather than creating new molecules from scratch.
The Bottom Line on Water Creation
So is it possible to make water? Absolutely. Chemistry gives us that power.
Is it practical, safe, or economically viable? Not remotely.
The explosive nature of hydrogen-oxygen reactions, the energy requirements, and the circular logic of sourcing hydrogen and oxygen from water itself all point to the same conclusion: making water isn’t the answer to water scarcity.
Real solutions already exist. Industrial water reuse technologies, advanced filtration systems, desalination improvements, and conservation efforts address water shortages without the risks of chemical synthesis.
Water covers most of our planet’s surface. The challenge isn’t quantity—it’s distribution, quality, and efficiency of use. Focus there, and the global water crisis becomes solvable without a single explosion.
