Quick Summary: Absolute zero, defined as 0 Kelvin (-273.15°C), represents the theoretical limit where atomic motion ceases and entropy reaches minimum. While researchers at institutions like MIT have cooled molecules to within 500 nanokelvin of absolute zero, the Third Law of Thermodynamics mathematically proves complete attainment is impossible. Each step closer requires exponentially more resources, making absolute zero an unreachable asymptote.
The quest to reach the coldest possible temperature has captivated physicists for over a century. But here’s the thing—despite achieving temperatures colder than interstellar space, scientists keep bumping into a fundamental barrier.
Absolute zero stands at 0 Kelvin, or -273.15 degrees Celsius. At this temperature, atomic motion theoretically reaches its minimum possible state. And while researchers have gotten remarkably close, the universe seems to have built-in rules that make complete attainment impossible.
This isn’t just some technical limitation we can engineer around. The impossibility of reaching absolute zero stems from the most fundamental laws of physics—laws that govern how energy, entropy, and temperature interact at quantum scales.
What Exactly Is Absolute Zero?
Absolute zero represents the lowest theoretical temperature limit, marking the point where particles possess minimal kinetic energy. This concept fundamentally underpins the Kelvin temperature scale, which Lord Kelvin helped establish in the 19th century.
At absolute zero, something fascinating happens. Entropy—the measure of disorder in a system—also reaches its minimum value. For a perfect crystalline structure, this entropy value would be precisely zero.
But atomic motion doesn’t completely stop. Quantum mechanics introduces zero-point energy, meaning particles retain residual motion even at the lowest possible temperatures. This quantum jitter prevents absolute stillness.
The Kelvin Scale and Temperature Measurement
The Kelvin scale starts at absolute zero and increases from there. Unlike Celsius or Fahrenheit, Kelvin contains no negative values—it begins at the fundamental limit and counts upward.
One Kelvin degree equals one degree Celsius in terms of temperature intervals. The difference? The Kelvin scale’s zero point sits at the absolute minimum, while Celsius arbitrarily sets zero at water’s freezing point.
According to NIST, Lord Kelvin’s contributions to temperature science established the metric unit we still use for precise scientific measurements. His work connected temperature to fundamental thermodynamic principles that govern energy transfer.
Why Can’t We Reach Absolute Zero?
The impossibility stems from the Third Law of Thermodynamics. This fundamental principle states that no process can reach absolute zero temperature in a finite number of steps within a finite time.
Research published in Nature in 2016 provides a rigorous mathematical derivation of this principle. The study quantifies exactly how many resources—time, energy, and steps—would be needed to cool a system to any given temperature.
Here’s what they found: the closer you get to absolute zero, the more resources you need. And that relationship isn’t linear—it’s exponential.
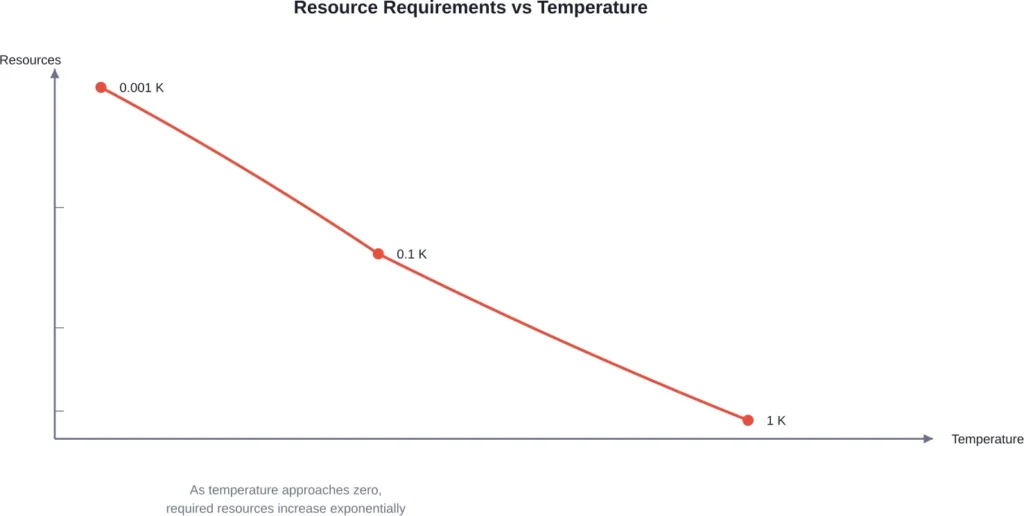
The Mathematics of Impossibility
The Nature study demonstrates that the obtainable temperature scales as an inverse power of cooling time. Translation? To halve the remaining distance to absolute zero, you need more than double the resources.
The minimal work needed grows proportionally to 1/T_min, where T_min represents the target minimum temperature. Approaching closer to zero doesn’t just get harder—it becomes prohibitively expensive in terms of energy and time.
This mathematical proof removes ambiguities that previously surrounded earlier derivations based purely on entropy. The unattainability principle now rests on solid quantitative foundations.
Quantum Mechanical Barriers
Quantum mechanics introduces another fundamental obstacle. Heisenberg’s uncertainty principle states that you cannot simultaneously know both a particle’s position and momentum with perfect precision.
Cooling requires measuring a system’s energy state to extract heat. But measurement itself introduces uncertainty. The more precisely you try to measure and extract energy, the more you disturb the system—adding energy back in.
Zero-point energy represents the minimum energy a quantum mechanical system can possess. Even at absolute zero, particles would retain this irreducible quantum motion. Perfect stillness violates quantum principles.
How Close Have Scientists Actually Gotten?
Despite theoretical impossibility, researchers have achieved astonishingly low temperatures. MIT physicists successfully cooled a gas of sodium potassium (NaK) molecules to a temperature of 500 nanokelvin—just half a millionth of a degree above absolute zero.
That’s over a million times colder than interstellar space. Outer space sits around 2.7 Kelvin due to cosmic microwave background radiation—practically tropical compared to laboratory achievements.
These extreme temperatures aren’t just record-breaking stunts. They enable observation of exotic quantum states like superconductivity and superfluidity that only emerge near absolute zero.
Advanced Cooling Techniques
Getting this cold requires sophisticated techniques that go far beyond conventional refrigeration. Scientists employ methods like laser cooling, evaporative cooling, and adiabatic demagnetization.
Laser cooling uses carefully tuned light to slow atoms. When photons interact with atoms at specific frequencies, they remove kinetic energy—essentially using light as a brake on atomic motion.
Evaporative cooling works similarly to how sweating cools your skin. The hottest particles escape from a magnetic trap, carrying away excess energy and leaving behind a colder sample.
Recent work at MIT (published January 2026) developed photonic chips that manipulate tightly focused, intersecting light beams to rapidly cool quantum computing systems. These advances push the boundaries of what’s achievable.
| Cooling Method | Typical Temperature Range | Primary Application |
|---|---|---|
| Liquid Nitrogen | 77 K (-196°C) | General laboratory cooling |
| Liquid Helium | 4.2 K (-269°C) | Superconducting magnets, MRI |
| Dilution Refrigeration | 0.002-0.01 K | Quantum computing, low-temp physics |
| Laser Cooling | Microkelvin range | Atomic clocks, Bose-Einstein condensates |
| Evaporative Cooling | Nanokelvin range | Ultracold quantum gases |
Bose-Einstein Condensates
At temperatures within a fraction of a degree from absolute zero, atoms can form Bose-Einstein condensates. In this state, thousands of atoms behave as a single quantum entity.
According to research on advances in laser-cooling technology, the path to creating these condensates was paved with ideas that initially seemed implausible. Early skeptics doubted laser cooling would work at all.
But it did work. And the achievement earned several physicists Nobel Prizes for developing techniques that became foundational to modern ultracold physics research.
NIST researchers routinely work with Bose-Einstein condensates to probe fundamental thermodynamic quantities. These quantum states enable precision measurements impossible at higher temperatures.
The Third Law of Thermodynamics Explained
The Third Law stands alongside the more famous First and Second Laws as a cornerstone of thermodynamics. While less intuitive than “energy is conserved” or “entropy increases,” it’s equally fundamental.
Two formulations exist: the unattainability principle and Nernst’s heat theorem. The unattainability principle directly states absolute zero cannot be reached. The heat theorem describes how entropy behaves as temperature approaches zero.
These formulations are connected but not identical. The 2016 Nature research clarified this relationship, showing how both versions emerge from the same underlying physics.
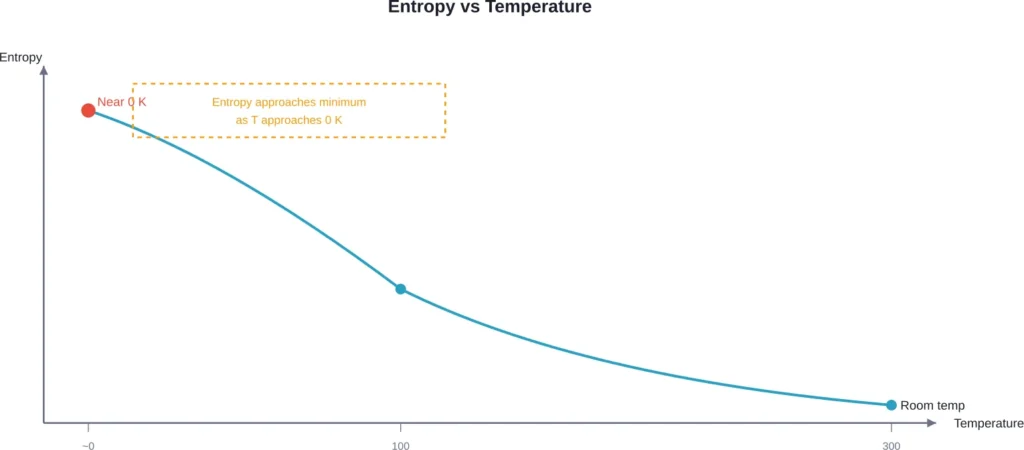
Entropy and the Approach to Zero
Entropy measures the number of possible microscopic configurations consistent with a system’s macroscopic state. More configurations mean higher entropy.
As temperature decreases, particles settle into lower energy states. Fewer configurations become accessible, and entropy drops. At absolute zero, a perfect crystal would have exactly one ground state configuration—zero entropy.
But reaching that singular configuration requires removing every last bit of thermal energy. Each step removes fewer and fewer energy quanta, requiring more precise control and more resources.
Why Perfect Crystals Matter
The Third Law specifically applies to perfect crystalline structures. Real materials contain defects, impurities, and structural irregularities that introduce residual entropy.
Even at extremely low temperatures, these imperfections prevent entropy from truly reaching zero. The crystal lattice retains multiple possible configurations due to defects.
This fact actually makes the unattainability principle even stronger. You’d need not just to cool a material to absolute zero, but also to create a perfectly ordered structure—both impossible tasks.
Practical Implications and Applications
The pursuit of temperatures near absolute zero isn’t purely academic. These extreme conditions reveal physics impossible to observe at normal temperatures.
Superconductivity—the complete absence of electrical resistance—only occurs below specific critical temperatures. For many materials, that threshold sits just a few degrees above absolute zero.
Research published by MIT physicists in 2008 examined superconducting compounds using scanning tunneling microscopes at ultracold temperatures. These investigations revealed characteristic patterns indicating charge density waves.
Quantum Computing and Information
Quantum computers require temperatures approaching absolute zero to function. Quantum bits (qubits) are extremely fragile, and thermal noise destroys quantum coherence.
Most quantum computing systems operate in dilution refrigerators at temperatures around 10-20 millikelvin. Even tiny temperature fluctuations introduce errors that corrupt calculations.
Research from Nature in 2016 connects the Third Law to fundamental limits on information processing. The work demonstrates that the speed at which information can be erased has ultimate bounds tied to cooling limitations.
Recent MIT developments in 2026 on chip-based trapped-ion quantum computers focus on efficient cooling methods. These advances could enable more stable quantum systems with better error correction.
Precision Measurement and Metrology
According to NIST physicist Stephen Eckel, ultracold atoms enable calibration-free measurements of thermodynamic quantities like pressure and temperature. His research uses atoms at both room temperature and ultracold conditions.
Atomic clocks—the most precise timekeeping devices—rely on laser-cooled atoms. Temperature affects atomic transition frequencies, so colder atoms produce more stable clock signals.
These precision measurements underpin GPS navigation, telecommunications networks, and fundamental physics experiments that test relativity and quantum mechanics.
The Curious Case of Negative Temperatures
Here’s where things get weird. Some theoretical frameworks discuss “negative absolute temperatures”—but these don’t mean colder than absolute zero.
In certain quantum systems with inverted energy populations, thermodynamic temperature can be defined as negative. However, MIT research from 2013 addressed the concept of negative absolute temperatures and perpetual motion machines, concluding both remain out of reach.
The concept relates to how entropy changes with energy in systems with bounded energy levels. When most particles occupy high-energy states—an inverted population—the mathematical definition of temperature becomes negative.
But these systems aren’t colder than absolute zero. They’re actually “hotter” than infinite temperature. The temperature scale wraps around: absolute zero, positive temperatures, infinite temperature, negative temperatures, back to absolute zero.
Real talk: this confuses even physicists. The key point is that negative temperatures don’t violate the Third Law. Absolute zero remains the unattainable lower bound for standard thermodynamic systems.
Comparing Absolute Zero to Cosmic Temperatures
Understanding absolute zero requires context. Where does it sit relative to temperatures found naturally in the universe?
Outer space has an ambient temperature of approximately 2.7 Kelvin—the temperature of cosmic microwave background radiation left over from the Big Bang. That’s about -270°C.
That might sound close to absolute zero. But remember, each degree Kelvin represents the same challenge. The difference between 2.7 K and 0 K is vast in terms of required cooling technology.
| Location/Phenomenon | Temperature (K) | Temperature (°C) |
|---|---|---|
| Absolute Zero (theoretical) | 0 | -273.15 |
| Coldest lab temperature (MIT, 2015) | 0.0000005 | -273.1499995 |
| Cosmic microwave background | 2.7 | -270.45 |
| Liquid helium boiling point | 4.2 | -268.95 |
| Liquid nitrogen boiling point | 77 | -196 |
| Room temperature | 293 | 20 |
| Sun’s surface | 5778 | 5505 |
The Coldest Known Place in the Universe
Human technology now regularly creates the coldest environments in the known universe. Laboratory achievements have exceeded the natural extremes found anywhere in space, a remarkable demonstration of experimental sophistication.
That’s remarkable when you think about it. Through laser cooling and magnetic trapping techniques developed over the past few decades, physicists have surpassed nature’s coldest conditions.
Common Misconceptions About Absolute Zero
Several myths persist about what absolute zero means and what would happen there. Let’s clear them up.
Myth: Atoms Stop Moving Completely
Not quite. Zero-point energy means particles retain quantum mechanical motion even at absolute zero. Electrons continue orbiting nuclei, molecules vibrate at their lowest energy levels.
Complete cessation of motion would violate Heisenberg’s uncertainty principle. If position were perfectly defined (a particle sitting still), momentum would become completely uncertain—physically impossible.
Myth: Time Stops at Absolute Zero
This science fiction trope doesn’t hold up. Time is a coordinate in spacetime, not dependent on temperature. Processes slow down dramatically as temperature drops, but time itself flows normally.
Chemical reactions essentially cease at temperatures near absolute zero. Atomic motion slows to quantum mechanical minimums. But temporal progression continues unchanged.
Myth: We’re Close to Reaching Absolute Zero
Scientists have achieved temperatures within billionths of a degree from absolute zero. Sounds close, right?
Wrong. The exponential resource requirements mean that last billionth of a degree is infinitely far away in practical terms. We’ll never close that final gap.
Think of it like Zeno’s paradox. You can halve the remaining distance infinitely many times, but you never actually arrive.

Theoretical Scenarios and Thought Experiments
What would happen if we somehow reached absolute zero? Entertaining this thought experiment reveals why the impossibility is actually necessary.
At true absolute zero, entropy would equal zero for a perfect crystal. The system would exist in a single, unique quantum state with no uncertainty.
But preparing a system in an exact quantum state requires infinite precision—another physical impossibility. Measurement and preparation processes introduce fundamental uncertainties.
Perpetual Motion Machines
MIT research from 2013 addressed the concept of negative absolute temperatures and perpetual motion machines, concluding both remain out of reach. A perpetual motion machine would violate the Second Law of Thermodynamics. Even if absolute zero were attainable, it wouldn’t circumvent fundamental energy conservation principles.
The notion that ultracold systems could somehow run indefinitely without energy input stems from misunderstanding what happens at low temperatures. Energy conservation still applies universally.
What About Heat Death of the Universe?
Some cosmological models predict the universe will eventually reach a state of maximum entropy—heat death. Would this represent a form of absolute zero?
Not really. Heat death means uniform temperature distribution, not absolute zero temperature. All available energy would be evenly spread out, making work impossible.
This equilibrium state would likely occur at a low but non-zero temperature. And even then, quantum fluctuations would persist, preventing true absolute zero conditions.
Recent Advances in Ultracold Physics
Research into temperatures approaching absolute zero continues advancing. New cooling techniques and quantum technologies emerge regularly.
Work on quantum thermodynamics published in Nature journals between 2016-2017 explored fundamental trade-offs between work extraction and fluctuations in quantum systems.
These studies found that for some processes like reversible cooling, fluctuations in work diverge. Realistic thermal machines cannot handle arbitrarily large fluctuations, which imposes practical limits beyond fundamental thermodynamic ones.
Chip-Based Quantum Systems
The January 2026 work at MIT on trapped-ion quantum computers demonstrates progress in miniaturizing ultracold systems. Photonic chips with precisely designed antennas manipulate intersecting light beams for rapid cooling.
These developments could enable more practical quantum computers that don’t require massive dilution refrigerators. Cooling efficiency directly impacts quantum computing viability.
But even with these advances, the fundamental limits remain unchanged. Engineering improvements bring us closer to absolute zero, but the Third Law ensures we never arrive.
Connections to Information Theory
Recent theoretical work connects thermodynamics to quantum information theory. The resources needed to cool a system relate to computational resources in surprising ways.
Research from 2016 established that cooling processes face similar restrictions to universal computers. The time and steps required to reach a given temperature follow computational complexity bounds.
This connection suggests deep links between thermodynamics, quantum mechanics, and information theory—three pillars of modern physics that increasingly appear as facets of a unified framework.
Frequently Asked Questions
The Third Law of Thermodynamics mathematically proves that reaching absolute zero would require infinite resources. Each step closer demands exponentially more energy and time. Additionally, quantum mechanics imposes zero-point energy, meaning particles retain residual motion even at the lowest possible temperatures. Both thermodynamic and quantum mechanical principles create insurmountable barriers.
MIT physicists successfully cooled a gas of sodium potassium (NaK) molecules to a temperature of 500 nanokelvin—just 0.0000005 degrees above absolute zero. This represents one of the coldest temperatures ever achieved and is over a million times colder than outer space. Various laboratories using laser cooling and evaporative cooling techniques routinely achieve temperatures in the microkelvin to nanokelvin range.
No, time would not stop at absolute zero. Time is a fundamental dimension of spacetime and doesn’t depend on temperature. While atomic motion would slow to its quantum mechanical minimum and chemical reactions would essentially cease, temporal progression would continue normally. This is a common science fiction misconception without basis in physics.
Atoms do not completely stop moving at absolute zero due to zero-point energy—the minimum energy required by quantum mechanics. Even at 0 Kelvin, particles would retain quantum mechanical motion. Complete cessation of all movement would violate Heisenberg’s uncertainty principle, which requires inherent uncertainty in position and momentum.
Outer space has an ambient temperature of approximately 2.7 Kelvin due to cosmic microwave background radiation. This is about -270.45°C, which seems close to absolute zero at -273.15°C. However, those final 2.7 degrees represent an enormous gap in terms of cooling difficulty. Laboratory techniques regularly achieve temperatures colder than any natural location in the known universe.
Quantum computing systems require temperatures around 10-20 millikelvin to maintain quantum coherence. Superconducting materials only exhibit zero electrical resistance below critical temperatures often just a few degrees above absolute zero. Precision atomic clocks, advanced physics research on Bose-Einstein condensates, and calibration-free measurement systems all depend on ultracold temperatures. These applications drive continued research into cooling technologies.
No technology, no matter how advanced, could reach absolute zero because the limitation is fundamental physics, not engineering. The Third Law of Thermodynamics represents a mathematical impossibility similar to traveling faster than light or creating energy from nothing. The exponential resource requirements mean the final approach to zero would require infinite time, energy, and cooling steps—physically impossible constraints that no technological advancement can overcome.
The Bottom Line on Absolute Zero
So, is it possible to reach absolute zero? The definitive answer is no.
This isn’t a limitation of current technology or a challenge to overcome with better engineering. The impossibility stems from fundamental laws governing how energy, entropy, and temperature interact at quantum scales.
The Third Law of Thermodynamics provides rigorous mathematical proof that any cooling process requires exponentially increasing resources as temperature approaches zero. The 2016 Nature research quantified these requirements explicitly, removing ambiguity from earlier formulations.
Quantum mechanics adds another layer of impossibility through zero-point energy and the uncertainty principle. Perfect stillness and zero entropy violate fundamental quantum constraints.
Yet the pursuit of ever-lower temperatures has driven remarkable discoveries. Superconductivity, superfluidity, Bose-Einstein condensates, and quantum computing all emerged from research at temperatures approaching absolute zero.
MIT researchers and other institutions continue pushing boundaries, achieving temperatures colder than anywhere in the natural universe. These accomplishments enable precision measurements, exotic quantum states, and technologies impossible at higher temperatures.
The universe seems to preserve absolute zero as an unreachable asymptote—something we can approach arbitrarily closely but never attain. Like the speed of light or Planck scale, it represents a fundamental boundary in the structure of physical law.
Understanding why absolute zero remains forever out of reach deepens our grasp of thermodynamics, quantum mechanics, and the elegant mathematical structure underlying reality. The impossibility itself teaches us about the nature of temperature, energy, and information.
Want to explore more about the frontiers of physics research? Check out current work on quantum computing, superconducting materials, and precision measurement techniques that all depend on temperatures approaching this fundamental limit.
