Quick Summary: Using Depo-Provera for more than 2 years raises concerns about bone mineral density loss and an increased risk of meningioma brain tumors. Medical research shows prolonged use correlates with higher tumor risk, and bone density may not fully recover after discontinuation. While Depo-Provera remains effective contraception, long-term users should discuss alternatives and monitoring with their healthcare provider.
Depo-Provera has been a popular birth control option since the FDA approved it in 1992. The contraceptive shot offers convenience—just four injections per year instead of daily pills. But what happens when those quarterly shots stretch beyond two years?
The answer involves more complexity than most patients realize when they first choose this method. Recent medical research and ongoing litigation have brought new attention to the long-term effects of extended Depo-Provera use.
Here’s what current evidence shows about using this birth control shot beyond the two-year mark.
Understanding Depo-Provera and How It Works
Depo-Provera is the brand name for depot medroxyprogesterone acetate (DMPA), an injectable contraceptive containing synthetic progesterone. The standard intramuscular formulation delivers 150 mg of medroxyprogesterone acetate every 12 weeks.
The medication works through several mechanisms. Primarily, it prevents ovulation by suppressing the release of eggs from the ovaries. It also thickens cervical mucus, creating a barrier that makes it harder for sperm to reach an egg.
Depo-Provera is 99 percent effective at preventing pregnancy when used correctly, with typical use effectiveness around 96%. This high efficacy rate made the shot particularly popular among teenage girls and women in their twenties who wanted reliable, long-acting contraception without daily adherence requirements.
By the time the FDA approved Depo-Provera in the United States, over 90 countries worldwide were already using it. The convenience factor drove widespread adoption—no daily pills to remember, no need for frequent prescription refills.
Why the Two-Year Threshold Matters
The two-year mark isn’t arbitrary. It represents a critical point where cumulative health effects become measurable and concerning.
Regulatory agencies haven’t pulled Depo-Provera from the market. But medical guidance increasingly emphasizes caution with extended use. The question isn’t whether Depo-Provera works as contraception—it does. The question is whether the long-term health trade-offs justify continued use beyond two years.
That calculation depends heavily on individual risk factors and alternative options.
Bone Mineral Density Loss: The Primary Concern
Bone health represents the most documented concern with long-term Depo-Provera use. The medication significantly impacts bone mineral density, particularly when used for extended periods.
According to research published in AJOG Global Reports, progestin-only contraceptives including DMPA show measurable effects on bone density during use. The rapid review examined evidence published between May 2012 and August 2023, analyzing studies on bone health effects of injectable contraceptives.
How Depo-Provera Affects Bones
Medroxyprogesterone acetate suppresses estrogen production. Lower estrogen levels interfere with normal bone remodeling, the continuous process where old bone tissue breaks down and new bone forms.
During Depo-Provera use, bone breakdown outpaces new bone formation. This imbalance leads to decreased bone mineral density over time.
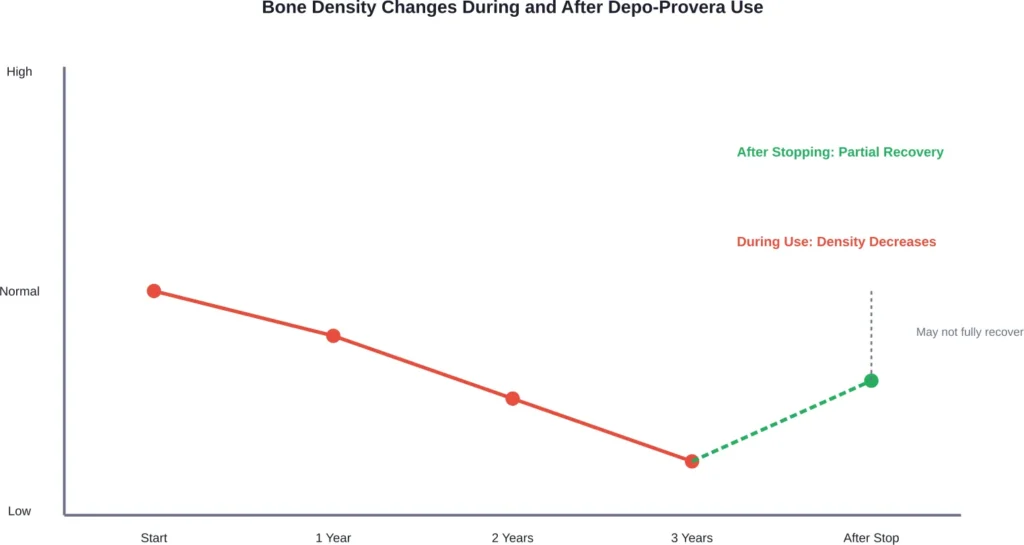
The loss becomes more pronounced the longer someone uses the medication. Women who use Depo-Provera for more than two years experience greater bone density reduction compared to shorter-term users.
Recovery After Discontinuation
Here’s where it gets complicated. Bone density may improve after stopping Depo-Provera, but recovery isn’t guaranteed to be complete.
Younger women tend to recover better than those approaching or past menopause. Adolescent users—a significant portion of Depo-Provera patients—face particular concerns because they’re still building peak bone mass during their teens and early twenties.
Some studies suggest bone density improves within 12 to 18 months after discontinuation. But whether bones fully return to pre-treatment density remains uncertain, especially for long-term users.
Recommendations for Bone Health
Medical sources recommend calcium supplementation of 1200 mg per day and vitamin D supplements for anyone using Depo-Provera. These supplements support bone health but don’t completely prevent bone density loss during use.
Weight-bearing exercise also helps maintain bone strength, though it can’t fully counteract the hormonal effects of the medication.
Meningioma Brain Tumors: The Emerging Risk
Recent research has identified a more alarming concern: intracranial meningiomas. These are tumors that develop in the protective membranes covering the brain and spinal cord.
The connection between Depo-Provera and meningiomas has sparked significant legal action and medical scrutiny in 2025 and 2026.
What Are Intracranial Meningiomas?
Meningiomas are typically slow-growing tumors arising from the meninges, the layers of tissue surrounding the brain and spinal cord. While most meningiomas are benign (non-cancerous), they can cause serious complications.
As these tumors grow, they compress brain tissue and nerves. This pressure leads to symptoms including headaches, vision problems, seizures, memory issues, and personality changes.
Treatment often requires surgical removal, radiation therapy, or long-term monitoring. Even benign meningiomas can significantly impact quality of life and require invasive interventions.
The Research Connection
Studies have found correlations between prolonged use of progestin-based medications and increased meningioma risk. The longer someone uses Depo-Provera, the higher their apparent risk becomes.
The biological mechanism involves progesterone receptors. Meningeal cells contain progesterone receptors, and exposure to synthetic progestins like medroxyprogesterone acetate may stimulate tumor growth in susceptible individuals.
Duration of use matters significantly. Short-term use appears to carry minimal risk, but multi-year exposure changes the calculation.
Warning Label Changes in Europe and the U.K.
Regulatory agencies in Europe and the United Kingdom have updated warning labels for progestin-based medications to include meningioma risk information. These changes reflect growing recognition of the connection between extended hormonal contraceptive use and brain tumor development.
The United States has been slower to implement similar label updates, though medical professionals are increasingly aware of the risk.
Other Long-Term Side Effects Beyond Two Years
Bone density loss and meningioma risk represent the most serious concerns. But prolonged Depo-Provera use can trigger additional effects worth considering.
Weight Changes
Many Depo-Provera users experience weight gain. The medication can induce changes in appetite, metabolism, and fat distribution. Weight changes may persist or worsen with continued use beyond two years.
Not everyone gains weight on Depo-Provera, but it’s common enough to warrant discussion with healthcare providers.
Menstrual Changes and Amenorrhea
Research published in Biological Reproduction examined how injectable contraceptives affect the hypothalamic-pituitary-gonadal axis. According to research on injectable contraceptives, only 11% of DMPA-IM users experienced normal menstrual patterns one year after initiation.
Amenorrhea—the complete absence of menstruation—becomes increasingly common with extended use. While some patients appreciate not having periods, prolonged amenorrhea can make it difficult to detect pregnancy if contraceptive failure occurs.
Two comparative studies showed less amenorrhea, bleeding, and spotting during the first six months of NET-EN use compared to DMPA. But amenorrhea incidence increases significantly with DMPA after the initial months.
Delayed Return to Fertility
According to research from the National Institutes of Health published in PMC, return to fertility after discontinuing subcutaneous depot medroxyprogesterone acetate varies considerably among individuals.
The contraceptive effects of the shot last for 14 weeks, although injections should be administered every 12 weeks to maintain consistent protection. After stopping, time to ovulation and fertility restoration can range from several months to over a year.
Women who’ve used Depo-Provera for multiple years may experience longer delays in fertility return compared to short-term users. Planning for pregnancy requires factoring in this potential delay.
Mood and Depression
Depo-Provera may worsen depression in susceptible individuals. Hormonal fluctuations affect neurotransmitter systems involved in mood regulation.
For someone with a history of depression, long-term use beyond two years could exacerbate symptoms. Mental health monitoring becomes important for extended users.
Headaches
Hormonal contraceptives can trigger or intensify headaches. Chronic headaches reduce quality of life and may indicate other issues requiring medical evaluation—particularly if meningioma risk is considered.
| Side Effect | Severity | Recovery After Stopping |
|---|---|---|
| Bone Density Loss | High | Partial, may not fully recover |
| Meningioma Risk | High | Risk persists; tumors require treatment |
| Weight Gain | Moderate | Variable, depends on lifestyle changes |
| Amenorrhea | Low to Moderate | Periods typically resume within months |
| Delayed Fertility | Moderate | Fertility returns but timing varies |
| Mood Changes | Moderate | Usually improves after discontinuation |
| Headaches | Low to Moderate | Often resolves after stopping |
What Happens After Stopping Depo-Provera
Discontinuing Depo-Provera after long-term use doesn’t immediately reverse all effects. The medication takes time to clear from the body, and some changes may be permanent.
Timeline for Medication Clearance
Each Depo-Provera injection provides contraceptive protection for approximately 14 weeks. After the last shot, medroxyprogesterone acetate gradually metabolizes over several months.
Full clearance typically takes six to nine months, though individual variation occurs based on metabolism, body composition, and duration of use.
Menstrual Cycle Return
Most women see menstrual periods resume within six to twelve months after their last injection. But some experience longer delays, particularly if they used Depo-Provera for several years.
Initial periods after discontinuation may be irregular in timing and flow. Cycle regularity typically improves over subsequent months.
Fertility Restoration
Time to ovulation after discontinuation varies. The narrative review published in PMC examined this specifically for subcutaneous depot medroxyprogesterone acetate.
Some women ovulate within a few months. Others wait a year or longer. Long-term users tend toward the longer end of this range.
For anyone planning pregnancy after Depo-Provera, consulting with a healthcare provider about realistic timelines proves valuable.
Bone Density Recovery
As mentioned earlier, bone density may improve after stopping but might not return to baseline, especially for multi-year users. Younger patients generally recover better than older ones.
Continued calcium and vitamin D supplementation, combined with weight-bearing exercise, supports bone health during the recovery period.
Current Lawsuits and Legal Considerations
The connection between Depo-Provera and meningioma brain tumors has triggered significant litigation. Law firms across the United States are filing lawsuits on behalf of women diagnosed with intracranial meningiomas after extended Depo-Provera use.
According to Sokolove Law, they have secured over $1.6 billion total for clients injured by dangerous drugs and medical devices. Depo-Provera cases represent a growing portion of pharmaceutical litigation.
Basis for Legal Claims
Lawsuits typically allege that manufacturers failed to adequately warn patients and healthcare providers about meningioma risks associated with long-term use. The argument centers on whether companies knew or should have known about tumor risks and whether they disclosed this information appropriately.
Updated warning labels in Europe and the U.K. strengthen these claims by demonstrating that regulatory agencies elsewhere recognized the risk warranting explicit warnings.
Who Can File a Lawsuit
Women diagnosed with intracranial meningiomas after using Depo-Provera for extended periods may have grounds for legal action. Particularly strong cases involve:
- Use beyond two years
- Meningioma diagnosis requiring surgery or radiation
- No prior family history of brain tumors
- Significant medical expenses or disability from the tumor
Consultation with attorneys specializing in pharmaceutical litigation provides case-specific guidance.
Potential Compensation
Successful claims may recover damages for medical expenses, lost wages, pain and suffering, and reduced quality of life. Settlement amounts vary based on individual circumstances, tumor severity, and treatment requirements.
Legal processes take time. But for someone facing substantial medical bills and long-term health consequences from a meningioma linked to Depo-Provera use, pursuing legal options makes sense.

Alternatives to Long-Term Depo-Provera Use
Given the concerns with extended Depo-Provera use, what alternatives exist for reliable contraception?
Other Hormonal Options
Birth control pills require daily adherence but allow users to stop immediately if side effects develop. Combined oral contraceptives contain both estrogen and progestin, while progestin-only pills avoid estrogen-related risks.
Hormonal intrauterine devices (IUDs) like Mirena or Skyla release progestin locally in the uterus. This localized delivery means lower systemic hormone exposure compared to injections. IUDs last three to seven years depending on the type.
Contraceptive implants like Nexplanon release progestin steadily for up to three years. They can be removed at any time if concerns arise.
Non-Hormonal Contraception
Copper IUDs provide highly effective contraception without any hormones. They work by creating an environment hostile to sperm and can remain in place for up to ten years.
Barrier methods like condoms, diaphragms, and cervical caps avoid hormonal effects entirely. Effectiveness depends on consistent correct use.
Permanent sterilization—tubal ligation for women or vasectomy for male partners—offers definitive contraception for those certain they don’t want future pregnancies.
Fertility Awareness Methods
Tracking ovulation through basal body temperature, cervical mucus monitoring, or fertility tracking apps allows contraception without devices or hormones. Effectiveness varies significantly based on diligence and cycle regularity.
These methods require commitment and education but appeal to individuals wanting to avoid medications entirely.
| Contraceptive Method | Effectiveness | Hormones | Duration |
|---|---|---|---|
| Depo-Provera | 96-99% | Progestin | 12 weeks per injection |
| Birth Control Pills | 91-99% | Estrogen + Progestin or Progestin-only | Daily |
| Hormonal IUD | 99% | Progestin (localized) | 3-7 years |
| Contraceptive Implant | 99% | Progestin | 3 years |
| Copper IUD | 99% | None | 10 years |
| Condoms | 85-98% | None | Per use |
| Sterilization | 99% | None | Permanent |
Having the Conversation with Healthcare Providers
If someone has been using Depo-Provera for approaching or exceeding two years, discussing the situation with a healthcare provider becomes important.
Questions to Ask
Here are key questions worth raising:
- What are my personal risk factors for bone density loss and meningiomas?
- Should I get a bone density scan to assess current bone health?
- What symptoms should prompt immediate medical evaluation?
- What alternative contraceptive methods might work for my situation?
- If I stop Depo-Provera, what’s the timeline for fertility return?
- Do I need specialized monitoring if I continue using the shot?
Informed Consent Matters
Real talk: patients deserve full information about long-term risks before continuing any medication beyond recommended durations. If a healthcare provider minimizes concerns or doesn’t discuss alternatives, seeking a second opinion makes sense.
Medical decisions work best when patients understand both benefits and risks clearly.
Who Should Avoid Long-Term Depo-Provera Use
Certain groups face elevated risks from extended Depo-Provera use:
Adolescents and young women: Those still building peak bone mass during their teens and early twenties risk interfering with optimal bone development. Bone density lost during these critical years may never fully recover.
Women with osteoporosis risk factors: Family history of osteoporosis, thin body frame, smoking, excessive alcohol use, or sedentary lifestyle all increase baseline bone health risks. Adding Depo-Provera compounds these concerns.
Anyone with depression history: Since Depo-Provera may worsen depression, those with mood disorders should carefully weigh risks versus benefits for long-term use.
Women planning pregnancy within two years: The delayed return to fertility makes Depo-Provera less ideal for anyone wanting to conceive relatively soon.
Those with personal or family history of brain tumors: Even a small meningioma risk becomes more concerning when baseline risk is already elevated.
Monitoring Recommendations for Current Users
For those who choose to continue Depo-Provera beyond two years despite the risks, certain monitoring steps reduce potential harm:
Bone density screening: Periodic DEXA scans measure bone mineral density objectively. Baseline scans before starting or early in use, followed by repeat scans every one to two years, track changes over time.
Calcium and vitamin D levels: Blood tests ensure supplementation achieves therapeutic levels.
Neurological symptom awareness: Any persistent headaches, vision changes, seizures, or cognitive changes warrant immediate medical evaluation, not just waiting for the next scheduled appointment.
Annual health reviews: Beyond routine gynecological care, comprehensive health assessments help catch developing issues early.
Weight and metabolic monitoring: Since weight gain is common, tracking weight, blood pressure, and metabolic markers helps identify concerning trends.
The Bottom Line on Using Depo-Provera Beyond Two Years
So what happens if you take Depo-Provera for more than two years?
The evidence points to real health concerns that shouldn’t be ignored. Bone mineral density decreases progressively with extended use, and recovery after stopping may be incomplete. The risk of developing intracranial meningiomas increases with duration of exposure, potentially requiring surgery and causing permanent neurological effects.
These aren’t hypothetical risks. They’re documented in medical research and reflected in regulatory warning label changes in multiple countries. Ongoing litigation underscores that manufacturers may not have adequately communicated these dangers to patients and healthcare providers.
But here’s the thing: individual circumstances vary. Someone with limited contraceptive options due to medical conditions, medication interactions, or access issues faces different calculations than someone with multiple viable alternatives.
The key is making informed choices based on complete information. Depo-Provera works effectively as birth control. The question isn’t whether it prevents pregnancy—it does. The question is whether the long-term health trade-offs justify continued use beyond two years for any particular individual.
For most patients, the answer leans toward finding alternatives. Numerous effective contraceptive methods exist with different risk profiles. Switching to options with fewer long-term concerns makes sense when feasible.
For those who continue Depo-Provera beyond two years, heightened monitoring and awareness of warning symptoms becomes essential. Don’t ignore persistent headaches or neurological symptoms. Get bone density assessed. Maintain calcium and vitamin D supplementation. Stay informed about new research findings.
And if diagnosed with a meningioma after extended Depo-Provera use, exploring legal options may provide compensation for medical expenses and suffering.
Frequently Asked Questions
Yes, extended Depo-Provera use can cause lasting effects. Bone mineral density loss may not fully recover after discontinuation, particularly in long-term users. The risk of developing intracranial meningiomas represents potentially permanent damage requiring surgical intervention. While some effects like menstrual changes and mood alterations typically improve after stopping, bone density and tumor risks carry long-term consequences.
Fertility return varies considerably among individuals. According to research from the National Institutes of Health, time to ovulation after discontinuing depot medroxyprogesterone acetate ranges from several months to over a year. Women who used Depo-Provera for multiple years often experience longer delays in fertility restoration compared to short-term users. Most women ovulate within 6 to 12 months after their last injection, but some wait longer.
Warning symptoms of intracranial meningiomas include persistent headaches, vision problems or vision loss, seizures, memory difficulties, personality changes, hearing loss or ringing in the ears, weakness in arms or legs, and balance problems. These symptoms develop as the tumor grows and compresses surrounding brain tissue and nerves. Anyone experiencing these symptoms after extended Depo-Provera use should seek immediate medical evaluation, including neurological imaging.
Yes, bone density screening makes sense for anyone who has used Depo-Provera for extended periods, particularly beyond two years. A DEXA scan objectively measures bone mineral density and identifies concerning loss before fractures occur. Baseline bone density assessment followed by periodic repeat scans tracks changes over time and helps guide decisions about continuing or discontinuing the medication.
Legal options exist for women diagnosed with intracranial meningiomas after extended Depo-Provera use. Lawsuits typically allege inadequate warnings about tumor risks associated with long-term use. According to legal sources, cases are strongest when use exceeded two years, meningioma diagnosis required surgery or radiation, and significant medical expenses or disability resulted. Consulting with attorneys specializing in pharmaceutical litigation provides case-specific guidance about potential claims.
Several alternatives offer highly effective contraception with different risk profiles. Copper IUDs provide 99% effectiveness without any hormones and last up to ten years. Hormonal IUDs deliver localized progestin with lower systemic exposure than injections. Contraceptive implants can be removed immediately if concerns arise, unlike injections that persist for months. The “safest” option depends on individual medical history, risk factors, and preferences. Discussing alternatives with a healthcare provider helps identify the best fit for specific circumstances.
Bone density typically improves somewhat after discontinuing Depo-Provera, but recovery may not be complete, especially for long-term users. Younger women generally recover better than those approaching menopause. Research suggests improvement occurs within 12 to 18 months after the last injection, but whether bones fully return to pre-treatment density remains uncertain. Continued calcium and vitamin D supplementation plus weight-bearing exercise supports bone health during recovery. Repeat bone density scans track actual recovery progress.
