Quick Summary: Drinking salt water, especially seawater, can be deadly to humans. Seawater contains roughly 3.5% salt, and when consumed, it forces your kidneys to work overtime trying to eliminate the excess sodium, ultimately causing severe dehydration, organ failure, and potentially death. While small amounts of diluted salt water (like sole water) may offer hydration benefits, seawater itself is never safe to drink.
One of the first survival lessons taught in school is simple: don’t drink seawater. But have you ever wondered what actually happens inside your body when you consume salt water?
The answer isn’t just “you get thirsty.” The physiological process that unfolds can lead to organ failure, seizures, and death. At the same time, some wellness trends promote drinking diluted salt water for health benefits.
So what’s the truth? Here’s everything that happens when salt water enters your system.
Why Seawater Is Deadly to Humans
Seawater contains salt. That’s obvious. What’s not obvious is just how much salt, and what that does to your cells.
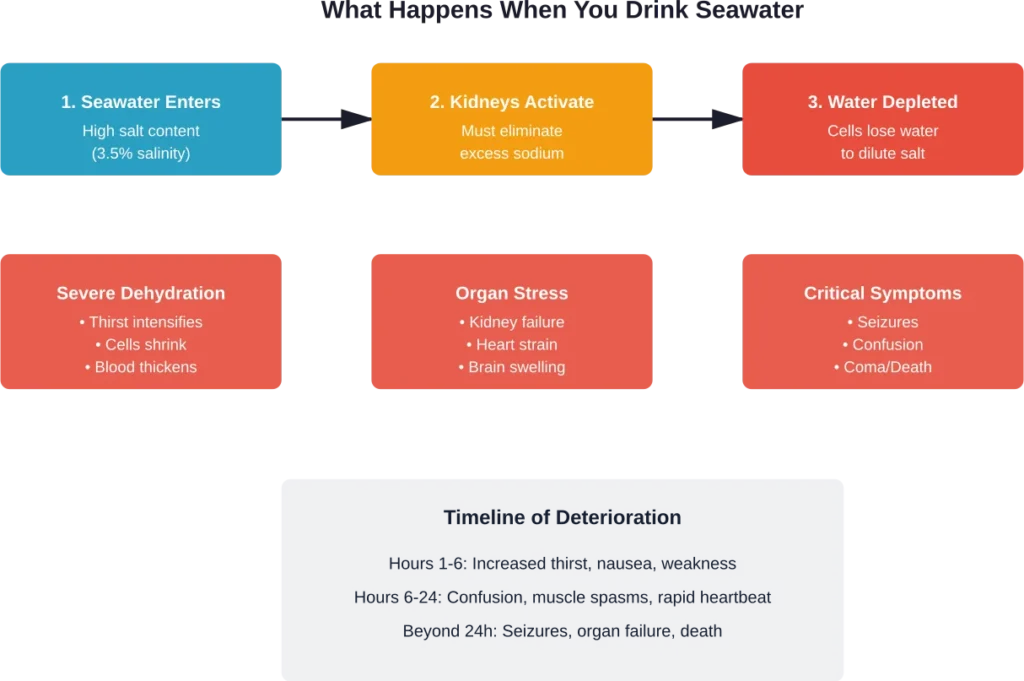
According to NOAA’s National Ocean Service, when humans drink seawater, their cells take in both water and salt simultaneously. The problem? Your kidneys can only produce urine that’s less salty than seawater.
The Kidney Overload Problem
Your kidneys are designed to filter out excess sodium and maintain electrolyte balance. But they need fresh water to do this job effectively.
When seawater enters your system, your kidneys must pull water from your body’s cells to dilute the excess salt for elimination. This creates a vicious cycle: drinking seawater actually removes more water from your body than it provides.
The result? Severe dehydration that worsens with every sip.
Hypernatremia: When Sodium Levels Spike
Medical research from the National Institutes of Health documents cases of severe hypernatremia — abnormally high sodium levels in the blood. In one documented case published in Electrolyte Blood Press, a patient who consumed excessive salt had serum sodium levels of 188.1 mmol/L (normal, 138–148 mmol/L).
This condition causes:
- Cellular dehydration throughout the body
- Brain swelling and neurological damage
- Muscle weakness and seizures
- Organ failure
- Death if untreated
According to research published in PMC, hypernatremia from salt poisoning is clinically rare but extremely dangerous. Standard care procedures involve aggressive rapid correction using controlled IV fluids, but even with treatment, outcomes can be fatal.
The Science Behind Salt Water and Your Cells
Understanding what happens requires looking at cellular biology.
Your cells maintain a delicate balance of water and electrolytes through a process called osmosis. Water naturally moves across cell membranes from areas of low salt concentration to areas of high salt concentration.
When you drink seawater, the salt concentration in your bloodstream skyrockets. Water then moves out of your cells to balance this concentration gradient. Your cells literally shrink.
Normal Sodium vs. Seawater Sodium
Here’s the comparison that explains why seawater is so dangerous:
| Measurement | Normal Blood Sodium | Seawater Sodium | Danger Threshold |
|---|---|---|---|
| Concentration (mmol/L) | 138-148 | ~470 | >160 |
| Salinity | 0.9% | 3.5% | >1.5% |
| Safe for consumption | Yes (naturally maintained) | No | No |
The gap between what your body maintains naturally and what seawater contains is massive. No amount of adaptation can make seawater safe to drink.
What About Small Amounts of Salt Water?
Not all salt water is seawater. The wellness community has embraced drinking diluted salt water, often called “sole water.”
This involves dissolving a small amount of sea salt or Himalayan salt in fresh water. The key difference? Concentration.
Sole Water vs. Seawater
Sole water typically contains less than 1% salt by volume — far below seawater’s 3.5%. At this concentration, the sodium content remains within safe limits for healthy individuals.
Some proponents claim sole water provides:
- Improved hydration through electrolyte balance
- Better mineral absorption
- Support for digestion
- Enhanced energy levels
But here’s the thing. Scientific evidence supporting these specific benefits remains limited.
According to the World Health Organization, the global mean intake of sodium for adults is 4310 mg/day (equivalent to 10.78 g/day salt) — more than double the WHO recommendation for adults of less than 2000 mg/day sodium (equivalent to < 5 g/day salt, or approximately one teaspoon). Most populations are consuming too much sodium, not too little.
The primary health effect associated with diets high in sodium is raised blood pressure, which increases the risk of cardiovascular disease.
When Diluted Salt Water Might Help
There are legitimate scenarios where controlled salt water consumption serves a purpose:
- Rehydration after extreme physical exertion with heavy sweating
- Recovery from severe vomiting or diarrhea causing electrolyte loss
- Medical conditions requiring sodium supplementation under doctor supervision
But these situations require precise sodium-to-water ratios, not random seawater consumption.

Medical Cases: Real-World Salt Poisoning
Medical literature contains sobering examples of what happens when people consume excessive salt.
Research published in Acute Medicine & Surgery describes a fatal case of acute hypernatremia resulting from massive intake of seasoning soy sauce. The patient’s serum sodium concentration reached 174 mEq/L initially, requiring aggressive fluid therapy.
Despite administering 1,600 mL of Ringer’s lactate solution in the first hour and continued correction with 5% dextrose, the outcome was fatal.
Another documented case involved a near-drowning victim who ingested seawater during a hurricane. According to research published in Western Journal of Medicine, severe hypernatremia from sea water ingestion presented immediate life-threatening complications requiring emergency intervention.
These cases highlight a critical point: once severe hypernatremia sets in, even aggressive medical treatment may not reverse the damage.
Why Your Kidneys Can’t Save You
Let’s address the elephant in the room. If kidneys are designed to filter salt, why can’t they just handle seawater?
The answer lies in basic physiology. According to MedlinePlus, kidneys need adequate fresh water to concentrate and eliminate waste products, including excess sodium.
When seawater enters your system, the kidneys must work to eliminate excess sodium. Your kidneys can only produce urine that is less salty than seawater, requiring more fresh water to dilute and excrete the salt load than the seawater initially provided.
The Concentration Problem
Seawater contains approximately 3.5% salt. Your kidneys must dilute excess sodium for elimination through urine.
Symptoms and Timeline of Salt Water Poisoning
What actually happens to someone who drinks seawater? The symptoms progress rapidly.
Initial Symptoms (First Few Hours)
- Intense thirst that worsens rather than improves
- Dry mouth and throat
- Nausea and vomiting
- Weakness and fatigue
- Headache
Progressive Symptoms (6-24 Hours)
- Confusion and disorientation
- Muscle spasms and cramps
- Rapid heartbeat
- Decreased urine output
- Extreme lethargy
Severe Symptoms (Beyond 24 Hours)
- Seizures
- Loss of consciousness
- Organ failure
- Coma
- Death
The timeline varies based on the amount consumed and individual health factors, but the progression is consistent and dangerous.
Emergency Treatment for Salt Water Ingestion
If someone accidentally consumes significant amounts of seawater, immediate medical attention is critical.
According to NIH research on hypernatremia treatment, the approach involves:
- Controlled administration of IV fluids (never rapid correction, which can cause brain damage)
- Careful monitoring of serum sodium levels
- Gradual sodium reduction using calculated fluid replacement
- Treatment of symptoms like seizures
- Monitoring for organ damage
The CDC emphasizes that healthcare professionals should be familiar with symptoms and management of diseases caused by toxins in seafood and saltwater exposure.
Never attempt to treat severe salt water poisoning at home. Correction must be gradual, with careful gradual sodium reduction, to prevent osmotic brain injury.
Who Should Avoid Salt Water Completely?
Even diluted salt water isn’t appropriate for everyone. Certain individuals should avoid it entirely:
| Condition | Risk Level | Recommendation |
|---|---|---|
| High blood pressure | High | Avoid all unnecessary sodium |
| Kidney disease | Very High | Strict medical supervision only |
| Heart disease | High | Follow cardiac diet restrictions |
| Pregnancy | Moderate | Consult obstetrician first |
| Diabetes | Moderate | Monitor sodium carefully |
| Children under 5 | High | More sensitive to sodium changes |
According to research on salt poisoning in children published in the British Medical Journal, infants are particularly vulnerable. The ratio of surface area to weight in infants weighing 5 kg is twice that of adults, and their skin is more permeable, meaning water loss is much greater.
Safe Alternatives for Hydration and Electrolytes
If electrolyte balance is the goal, safer alternatives exist:
- Coconut water: Natural electrolytes without excessive sodium
- Oral rehydration solutions: Medically formulated electrolyte balance
- Sports drinks: Controlled sodium and electrolyte content
- Mineral water: Natural minerals without dangerous salt levels
- Electrolyte tablets: Precise dosing for athletic recovery
These options provide hydration benefits without the extreme risks associated with seawater or even heavily salted water.
The Bottom Line on Drinking Salt Water
Here’s what matters most.
Seawater is never safe to drink. The salt concentration is too high for human kidneys to process without causing net water loss and potentially fatal hypernatremia.
Diluted salt water (sole water) may offer minor benefits for some healthy individuals, but scientific evidence supporting specific health claims remains limited. Most people already consume more than enough sodium through their regular diet.
If hydration and electrolyte balance are concerns, medically formulated solutions provide safer, more effective options.
The old survival advice remains absolutely correct: when stranded without fresh water, never drink seawater. It will accelerate dehydration and death rather than preventing it.
Always prioritize finding fresh water sources or using proper desalination methods before considering any saltwater consumption.
Frequently Asked Questions
Yes, even small amounts of seawater can cause problems. While a single small sip likely won’t cause immediate harm to a healthy adult, repeated consumption quickly leads to dehydration and elevated sodium levels. The kidneys must use more water than the seawater provides to eliminate the excess salt, creating a net water loss. There’s no safe amount of seawater for regular consumption.
The fatal dose varies by individual body weight, health status, and hydration level, but medical research shows that consuming quantities that raise serum sodium above 160 mmol/L creates life-threatening conditions. For an average adult, drinking several cups of seawater without access to fresh water could be fatal within days. Children are far more sensitive and can experience dangerous sodium levels with smaller amounts.
Swallowing small amounts while swimming is generally not dangerous for healthy individuals. Rinse your mouth with fresh water when possible and drink fresh water to help dilute any salt consumed. If someone consumes large quantities or shows symptoms like confusion, severe thirst, vomiting, or weakness, seek immediate medical attention. Do not try to induce vomiting, as this worsens dehydration.
Scientific evidence for specific sole water benefits remains limited. While diluted salt water provides sodium and trace minerals, most people already consume more sodium than the WHO recommends (adults average 4,310 mg/day versus the recommended maximum of 2,000 mg/day). Sole water may help individuals with specific electrolyte deficiencies or those recovering from illness causing sodium loss, but it’s not necessary for most people and could be harmful for those with hypertension or kidney disease.
No. Human kidneys cannot adapt to process seawater safely. Unlike some marine animals with specialized salt glands, humans lack the biological mechanisms to eliminate highly concentrated salt without using more water than the seawater provides. Evolution has not equipped humans to drink seawater, and no amount of gradual exposure will change this fundamental physiological limitation.
Some survival manuals suggest mixing small amounts of seawater with fresh water to extend fresh water supplies, but this is controversial and risky. The practice only works if the ratio keeps total salt concentration well below 1%, and it still accelerates dehydration compared to drinking fresh water alone. Most survival experts recommend avoiding this practice entirely and instead focusing on finding fresh water, collecting rain, or using proper desalination methods.
The critical difference is concentration. Ocean water contains approximately 3.5% salt by weight, while sole water or salted drinking water typically contains less than 1% salt. This concentration difference is everything — the lower concentration allows kidneys to process and eliminate the sodium without requiring more water than consumed. Additionally, some sea salts contain trace minerals that regular table salt lacks, though the health significance of these minerals in such small quantities is debatable.
