Quick Summary: Using expired semaglutide may result in reduced effectiveness rather than immediate danger. The medication can lose potency over time, potentially leading to poor blood sugar control or diminished weight loss results. While expired semaglutide isn’t typically toxic, healthcare providers recommend against using it due to unpredictable therapeutic outcomes.
Semaglutide has become one of the most talked-about medications for type 2 diabetes and weight loss. Whether it’s Ozempic, Wegovy, or a compounded version, millions rely on this GLP-1 receptor agonist daily.
But what happens when the expiration date passes?
Maybe the pharmacy delivered a new supply late. Perhaps you found an old pen in the back of your fridge. Or you’re using compounded semaglutide and aren’t sure about its beyond-use date.
Here’s the thing though—expired semaglutide behaves differently than many other medications. Understanding what changes after expiration can help determine the best course of action.
Understanding Semaglutide Expiration vs. Beyond-Use Dates
Not all expiration dates are created equal.
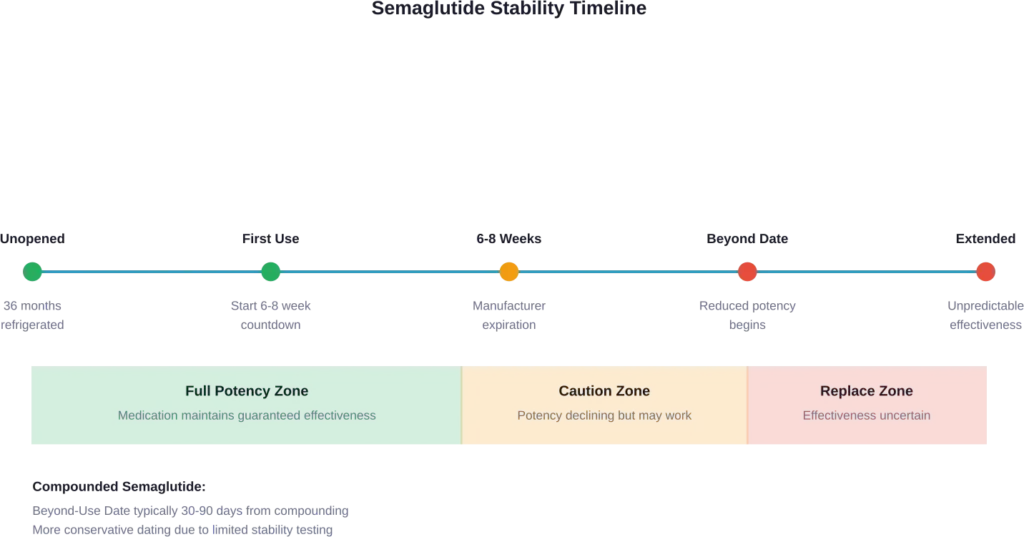
Brand-name semaglutide products like Ozempic and Wegovy come with FDA-approved expiration dates. These indicate how long the manufacturer guarantees full potency when stored correctly. Semaglutide is stable for 36 months when refrigerated and unopened.
Once opened, the timeline changes dramatically. Most semaglutide pens remain effective for 6-8 weeks after the first use.
Compounded semaglutide operates under different rules. These formulations don’t carry FDA approval and instead use beyond-use dates (BUDs) assigned by the compounding pharmacy. The BUD is typically much shorter than brand-name expiration dates—often 30-90 days depending on the formulation and storage conditions.
Why the difference? Compounded medications lack the extensive stability testing that brand-name drugs undergo. Pharmacies must be conservative with their dating to ensure patient safety.
What Happens to Semaglutide After It Expires?
The primary concern with expired semaglutide is reduced potency, not toxicity.
Semaglutide is a biologic medication—a complex protein-based molecule that can degrade over time. According to NIH research, semaglutide works by decreasing hunger, increasing satiety, and delaying gastric emptying, and weight loss can result in a 10-15% decline in body weight.
When the medication degrades, these effects diminish.
The molecular structure can break down through several mechanisms. Temperature fluctuations accelerate degradation. Exposure to light can damage the peptide chains. Even normal aging causes some molecular breakdown.
But does expired semaglutide become dangerous?
Real talk: There’s no evidence that expired semaglutide produces toxic byproducts. The medication simply becomes less effective. Think of it like old vitamins—not harmful, just not doing the job anymore.
Potential Risks and Safety Concerns
While expired semaglutide isn’t typically toxic, using it carries real risks.
Therapeutic Failure
The most significant risk is treatment failure. For patients with type 2 diabetes, this means blood glucose levels may rise above target ranges. Poor glycemic control over time increases the risk of diabetes complications including neuropathy, retinopathy, and cardiovascular issues.
For weight management patients, expired medication may provide little to no appetite suppression. The return of hunger and reduced satiety can derail progress and cause frustration.
Unpredictable Dosing
Here’s where it gets tricky. Degraded semaglutide doesn’t lose potency uniformly. One dose might retain 80% effectiveness while another drops to 40%.
This unpredictability makes it impossible to adjust dosing reliably. Patients can’t simply “take more” to compensate because the degradation rate is unknown.
Gastrointestinal Side Effects
According to research published in the Journal of Clinical Medicine, the most frequent adverse events associated with GLP-1 receptor agonists are gastrointestinal in nature, with GI adverse events developing in 40-70% of treated patients—namely nausea, vomiting, diarrhea, and constipation.
Degraded medication could potentially alter how the body processes these compounds, though there’s limited research on this specific scenario.
Metabolic Disruption
Patients who’ve achieved stable blood sugar control or weight maintenance may experience sudden metabolic changes when their medication loses effectiveness. This can be particularly concerning for diabetes patients who’ve adjusted other medications or dietary habits based on semaglutide’s effects.
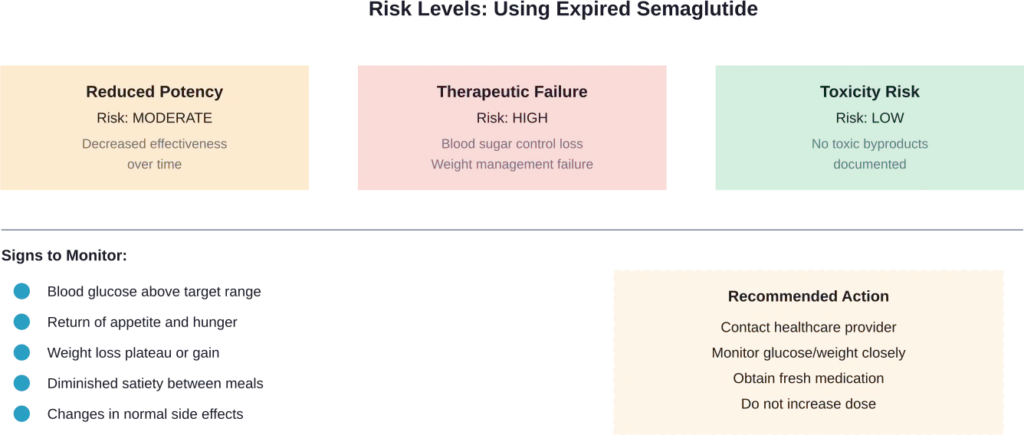
Signs You Should Contact Your Healthcare Provider
Certain symptoms warrant immediate medical attention.
Contact a healthcare provider if experiencing:
- Blood glucose readings consistently above target range (for diabetes patients)
- Rapid weight regain after stable maintenance
- Sudden return of intense hunger or food cravings
- Unusual gastrointestinal symptoms different from typical semaglutide side effects
- Signs of hyperglycemia including excessive thirst, frequent urination, or fatigue
These symptoms suggest the medication is no longer providing therapeutic benefits.
Some patients mistakenly assume they’ve developed tolerance to semaglutide. But wait. True tolerance to GLP-1 receptor agonists is relatively rare. More often, apparent tolerance stems from expired or improperly stored medication.
How Storage Affects Semaglutide Stability
Proper storage dramatically impacts how long semaglutide remains effective.
Temperature Requirements
Unopened semaglutide must be refrigerated at 36-46°F (2-8°C). This temperature range preserves the protein structure and maintains potency for up to 36 months.
Once opened, brand-name pens can be kept at room temperature (below 86°F/30°C) for 6-8 weeks. However, continued refrigeration often extends effectiveness slightly.
Compounded semaglutide typically requires continuous refrigeration both before and after opening. Check the pharmacy’s specific storage instructions.
Critical Storage Mistakes
Certain conditions accelerate degradation:
| Storage Error | Impact on Medication | Time to Degradation |
|---|---|---|
| Freezing | Protein structure damage | Immediate—discard if frozen |
| Direct sunlight exposure | Accelerated molecular breakdown | Hours to days |
| Temperatures above 86°F | Rapid potency loss | Days to weeks |
| Repeated temperature changes | Cumulative degradation | Weeks to months |
| Leaving pen uncapped | Exposure to air and light | Variable |
Refrigerator Placement Matters
Not all spots in the fridge are equal. Store semaglutide in the main compartment, never in the door. Door storage exposes medication to temperature fluctuations every time the fridge opens.
Avoid placing pens near the back wall where temperatures can drop near freezing. The middle shelf toward the front provides the most stable environment.
Compounded Semaglutide: Special Considerations
Compounded semaglutide presents unique challenges.
These formulations don’t undergo the same rigorous stability testing as FDA-approved products. Compounding pharmacies prepare semaglutide by reconstituting pharmaceutical-grade ingredients, but the final product’s stability can vary based on numerous factors.
The beyond-use date assigned to compounded semaglutide is inherently conservative. Pharmacies must follow USP guidelines that prioritize safety over maximizing shelf life.
Sound familiar? Many patients report receiving compounded semaglutide with BUDs of just 30-45 days. This short window can create challenges for those who order in bulk or experience shipping delays.
Research published in medical journals shows that compounded medications may have different stability profiles than their brand-name counterparts due to variations in excipients, pH buffering, and preservative systems.
When Compounded Semaglutide Reaches Its BUD
The beyond-use date represents the pharmacy’s guarantee of potency. After this date, the compounding pharmacy can no longer vouch for the medication’s effectiveness.
This doesn’t mean the medication instantly becomes ineffective. However, the rate of degradation accelerates and becomes unpredictable.
For patients using compounded semaglutide, adhering strictly to the BUD is particularly important because there’s less data on long-term stability compared to brand-name products.
What Healthcare Providers Recommend
Medical professionals consistently advise against using expired semaglutide.
The primary reason isn’t safety—it’s predictability. Healthcare providers need to know that prescribed treatments deliver consistent therapeutic effects. Expired medication introduces too many variables.
For diabetes management, unreliable medication can undermine carefully calibrated treatment plans. Providers may have adjusted insulin doses, meal planning, or other diabetes medications based on semaglutide’s effects. When that foundation becomes unstable, the entire treatment approach needs reassessment.
Weight management patients face different challenges. According to NIH research, weight loss can be profound with semaglutide, resulting in a 10-15% decline in body weight. When the medication loses potency, patients may interpret weight plateaus or regain as personal failure rather than medication degradation.
The Cost-Effectiveness Question
Now, this is where it gets interesting. Some patients consider using expired semaglutide due to cost concerns. The medication can be expensive, particularly without insurance coverage.
Healthcare providers acknowledge this reality but point out that ineffective medication wastes money regardless of the purchase price. Paying for a dose that doesn’t work provides no value.
Better approaches include:
- Discussing generic alternatives or different GLP-1 receptor agonists with better coverage
- Exploring patient assistance programs offered by manufacturers
- Investigating compounding pharmacy options with better pricing
- Asking about different dosing schedules that might reduce monthly costs
What to Do If You Only Have Expired Semaglutide
Real-world situations don’t always align with ideal recommendations.
If expired semaglutide is the only option temporarily, certain precautions can minimize risks:
For diabetes patients: Increase blood glucose monitoring frequency. Check levels before meals and two hours after eating to detect any changes in glycemic control early. Keep rapid-acting glucose available in case readings become elevated.
For weight management patients: Track weight daily and monitor appetite levels closely. Document any changes in hunger patterns, satiety, or food cravings. This data helps healthcare providers assess whether the medication is still providing benefits.
For all patients: Contact the prescribing provider immediately to explain the situation. Don’t wait until the next scheduled appointment. Many providers can expedite prescription transfers or find alternative solutions.
Document the medication’s expiration date and storage history. This information helps providers understand the likely degree of degradation.
Never Double the Dose
Some patients consider increasing the dose to compensate for reduced potency. This is dangerous.
The degree of degradation is unknown. Doubling a dose that’s retained 70% potency could result in a 1.4x overdose rather than the intended compensation. Semaglutide overdose can cause severe nausea, vomiting, and dangerous drops in blood sugar.
Proper Disposal of Expired Semaglutide
Once expired medication is replaced, proper disposal protects both people and the environment.
Never flush semaglutide down the toilet or throw it in regular trash. Pharmaceutical compounds can contaminate water supplies and pose risks to others who might access discarded medication.
The best disposal method involves FDA-approved medication take-back programs. Many pharmacies, hospitals, and law enforcement agencies operate permanent collection sites. These programs ensure safe disposal through controlled incineration.
Drug take-back events occur periodically in most communities. The DEA sponsors National Prescription Drug Take-Back Days twice yearly, typically in April and October.
If no take-back option is available, the FDA recommends mixing medication with undesirable substances like dirt, cat litter, or coffee grounds in a sealed bag before placing it in household trash. Remove any personal information from the medication packaging first.
How to Prevent Expiration Issues
Prevention beats problem-solving.
Several strategies help ensure medication remains fresh:
Order strategically: Calculate exactly how much medication is needed before the expiration date or BUD. For weekly semaglutide injections, don’t order more than an eight-week supply at once for brand-name products. For compounded versions, match orders to the specific BUD provided.
Track dates actively: Mark expiration dates on a calendar with reminders set one week prior. This provides time to order replacements before running out.
Inspect upon delivery: Check expiration dates immediately when medication arrives. Pharmacies occasionally ship products with short dating. Addressing issues immediately while return windows are open prevents problems later.
Rotate stock: If maintaining backup supplies, use the first-in-first-out principle. Move older medication to the front of the refrigerator and newer supplies to the back.
Monitor storage conditions: Consider placing a refrigerator thermometer near medication storage to ensure temperatures remain in the proper range. Some pharmacies provide temperature monitoring devices with expensive medications.
| Medication Type | Unopened Stability | After First Use | Key Storage Notes |
|---|---|---|---|
| Ozempic (brand) | 36 months refrigerated | 56 days | Can be room temp after opening |
| Wegovy (brand) | 36 months refrigerated | 56 days | Can be room temp after opening |
| Compounded semaglutide | Per BUD (30-90 days typical) | Per BUD | Continuous refrigeration required |
| Rybelsus (oral) | 36 months at room temp | N/A (individual tablets) | Protect from moisture |
The Science Behind GLP-1 Receptor Agonist Degradation
Understanding why semaglutide degrades requires looking at its molecular structure.
Semaglutide is a glucagon-like peptide-1 receptor agonist—a synthetic version of a naturally occurring hormone. According to NIH research, it’s a recombinant DNA-produced polypeptide analogue of human GLP-1.
The molecule consists of 31 amino acids arranged in a specific sequence. This protein structure gives semaglutide its therapeutic properties but also makes it vulnerable to degradation.
Several processes can damage the molecule:
Hydrolysis: Water molecules can break peptide bonds between amino acids. This process occurs slowly at proper storage temperatures but accelerates with heat.
Oxidation: Certain amino acids like methionine are susceptible to oxidation, particularly when exposed to light or oxygen. Oxidized amino acids lose their original function.
Aggregation: Individual semaglutide molecules can clump together, forming aggregates that don’t work properly. Temperature fluctuations promote aggregation.
Deamidation: Amino acids like asparagine and glutamine can lose their amino groups over time, altering the molecule’s structure and function.
Manufacturers incorporate stabilizers and preservatives to slow these processes, but degradation remains inevitable over extended periods.
Emerging Research on GLP-1 Medications
The field of GLP-1 receptor agonists continues evolving rapidly.
Recent research published in medical journals examines novel GLP-1-based medications for type 2 diabetes and obesity. Multireceptor agonists and oral GLP-1 therapies may reshape the future landscape by offering more effective and better-tolerated options.
Research from the EudraVigilance database analyzed real-world semaglutide safety data. According to the World Health Organization, in 2022, 2.5 billion adults were overweight, and of them, 890 million were obese (~12.5%).
Studies document the most common adverse events. Research shows that gastrointestinal issues remain the most frequent side effects, with reported associations including headache (ROR 1.74) and migraine (ROR 1.28), though neuropsychiatric adverse events have also been reported in pharmacovigilance databases.
But wait. There’s limited research specifically examining expired semaglutide’s effects. Most studies focus on properly stored medication within its approved dating.
Alternative Approaches During Medication Gaps
When semaglutide expires and replacement isn’t immediately available, other strategies can help maintain progress.
For diabetes management, focusing on dietary carbohydrate control becomes crucial. Reducing carbohydrate intake lowers glucose levels independently of medication. Increasing physical activity also improves insulin sensitivity.
Weight management patients can emphasize behavioral strategies that semaglutide supports. The medication reduces hunger, but it doesn’t create the caloric deficit—eating less does. Mindful eating practices, smaller portions, and protein-rich meals can help maintain appetite control.
These approaches aren’t permanent solutions. Semaglutide provides therapeutic effects that diet and exercise alone can’t replicate for most patients. However, they can bridge short gaps between medication supplies.
Insurance and Prescription Considerations
Many patients don’t realize that insurance companies often allow early refills for refrigerated medications nearing expiration.
Contact the insurance provider to explain that current medication will expire before the standard refill date. Many plans make exceptions for temperature-sensitive biologics.
Similarly, pharmacies can sometimes request override authorizations for early fills when expiration dates create access issues. The prescribing provider may need to document medical necessity.
For patients using compounded semaglutide, comparing beyond-use dates across different compounding pharmacies can reveal significant variation. Some pharmacies use more conservative dating than necessary. Switching to a pharmacy that provides longer BUDs (while still following proper guidelines) can reduce waste.
Frequently Asked Questions
Using semaglutide shortly after expiration carries minimal risk of toxicity but may result in reduced effectiveness. The medication doesn’t suddenly become dangerous at midnight on the expiration date. However, potency begins declining, and the rate of degradation becomes unpredictable. For best results, obtain fresh medication rather than using expired doses. If using recently expired semaglutide is unavoidable, monitor blood glucose or weight closely for signs of reduced effectiveness.
Visual inspection provides clues. Semaglutide should be clear and colorless. Discard the medication if it appears cloudy, discolored, contains particles, or has changed consistency. These visible changes indicate degradation or contamination. However, degraded semaglutide may still look normal—reduced potency isn’t always visible. Always check expiration dates regardless of appearance.
Expiration dates on FDA-approved brand-name medications indicate how long manufacturers guarantee full potency based on extensive stability testing. Beyond-use dates (BUDs) on compounded medications represent conservative estimates assigned by compounding pharmacies following regulatory guidelines. BUDs are typically much shorter because compounded medications undergo less stability testing. Both dates indicate when potency can no longer be guaranteed.
Expired semaglutide is unlikely to cause illness or toxicity. The primary concern is reduced effectiveness rather than safety. No evidence suggests that degraded semaglutide produces harmful byproducts. However, therapeutic failure from ineffective medication can lead to health consequences—elevated blood sugar in diabetes patients or weight regain in those using it for obesity management. These secondary effects pose the real health risks.
Never freeze semaglutide. Freezing damages the protein structure permanently, rendering the medication ineffective. If semaglutide accidentally freezes, discard it even if it later thaws and appears normal. The molecular damage from freezing is irreversible. Store semaglutide in the main refrigerator compartment away from areas that might reach freezing temperatures.
Compounded semaglutide has shorter beyond-use dates (typically 30-90 days) compared to brand-name products (36 months unopened). This doesn’t necessarily mean compounded versions degrade faster—rather, they lack the extensive stability data that supports longer dating for FDA-approved products. Compounding pharmacies must use conservative dating to ensure patient safety in the absence of comprehensive testing.
Monitor for signs of reduced effectiveness rather than immediate danger. For diabetes patients, check blood glucose more frequently over the next several days. For weight management, pay attention to appetite and satiety levels. Contact the healthcare provider to report the incident and request replacement medication. Document when the medication expired and how it was stored. Most importantly, don’t panic—single doses of recently expired semaglutide rarely cause acute problems.
Conclusion: Prioritizing Medication Effectiveness and Safety
Using expired semaglutide represents an effectiveness problem more than a safety crisis.
The medication won’t suddenly become toxic, but it can’t be relied upon to deliver consistent therapeutic results. For diabetes management, this unpredictability undermines treatment plans and puts glycemic control at risk. For weight management, reduced potency can stall progress and create frustration.
Healthcare providers universally recommend against using expired medications because treatment success depends on predictable, reliable therapeutic effects. The potential cost savings from using expired medication disappear when those doses fail to work.
Proper storage extends semaglutide’s useful life. Maintaining refrigeration at 36-46°F, protecting medication from light, avoiding temperature fluctuations, and never freezing the product all help preserve potency through the expiration date.
When medication does expire, proper disposal protects the environment and prevents accidental exposure. FDA-approved take-back programs provide the safest disposal method.
The key takeaway? Treat expiration dates as firm boundaries, not suggestions. Semaglutide’s therapeutic benefits depend on maintaining full potency. Using expired medication introduces too many variables and too much risk for too little benefit.
Contact a healthcare provider immediately if currently using or considering using expired semaglutide. Multiple solutions exist—from early refill authorizations to alternative medications—that don’t compromise treatment effectiveness. The investment in fresh medication pays dividends in consistent results and peace of mind.
