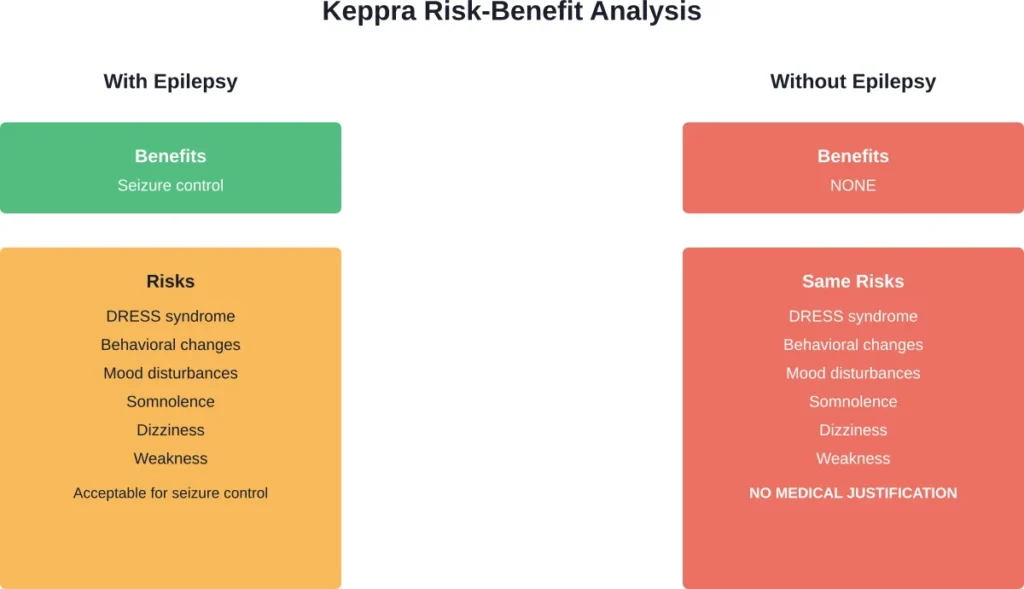
Quick Summary: Taking Keppra (levetiracetam) without a diagnosed seizure disorder is medically inappropriate and can expose individuals to serious side effects without providing therapeutic benefit. The FDA warns that Keppra can cause rare but life-threatening reactions like DRESS syndrome, along with common side effects including behavioral changes, mood disturbances, and somnolence. If prescribed Keppra despite not having epilepsy, consult a healthcare provider immediately to discuss safer alternatives.
Keppra, known generically as levetiracetam, ranks among the most widely prescribed antiseizure medications worldwide. But what happens when someone takes this powerful drug without actually having epilepsy or a diagnosed seizure disorder?
The short answer? Nothing good.
Taking Keppra without medical necessity exposes the body to significant risks without any therapeutic benefit. This isn’t about casual side effects—the FDA has issued specific warnings about life-threatening reactions that can occur with this medication.
Understanding What Keppra Actually Does
Levetiracetam works by binding to a protein called SV2A in the brain. According to the National Institutes of Health, this protein is part of secretory vesicle membranes that mediate calcium-dependent vesicular neurotransmitter release. The binding of levetiracetam to SV2A appears to decrease the rate of vesicle release.
In simpler terms, Keppra slows down the electrical signals in the brain that can trigger seizures. For someone with epilepsy, this mechanism prevents the bursts of abnormal electrical activity that characterize seizures.
But here’s the thing—if the brain isn’t experiencing abnormal electrical activity to begin with, Keppra is essentially disrupting normal neurotransmitter function without cause.
The NHS explains that seizures are bursts of electrical activity in the brain that temporarily affect how it works. Levetiracetam slows these electrical signals down to stop seizures. The exact way this medication works is not fully understood, which makes unnecessary use even more problematic.
The FDA’s Serious Warning About Keppra
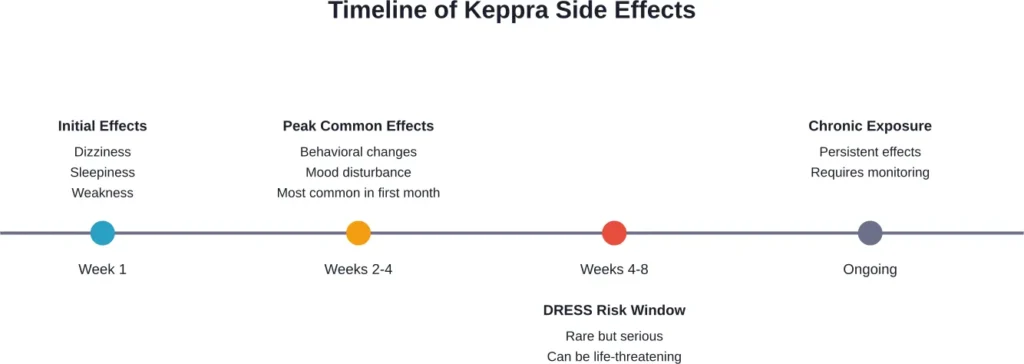
FDA issued a Drug Safety Communication on 11-28-2023 warning about a rare but serious reaction associated with levetiracetam (Keppra, Keppra XR, Elepsia XR, Spritam) and clobazam (Onfi, Sympazan). This reaction is called Drug Reaction with Eosinophilia and Systemic Symptoms, or DRESS.
DRESS can be life-threatening if not diagnosed and treated quickly.
According to the FDA warning, this reaction may start as a rash but can quickly progress to affect multiple organ systems. The risk exists for anyone taking Keppra—whether they have epilepsy or not. But taking the medication without medical necessity means accepting this risk with zero therapeutic benefit in return.
The FDA specifically warns that DRESS associated with levetiracetam requires immediate medical attention. Symptoms can include fever, rash, swollen lymph nodes, and signs of organ involvement.
Common Side Effects When Taking Keppra
A systematic review published in PLoS One examined the safety of levetiracetam in pediatric patients. The research identified a total of 1,913 adverse events reported across studies. The most common adverse events were behavioral problems and somnolence, which accounted for 10.9% and 8.4% of all adverse events respectively in prospective studies. Among 21 prospective studies involving 1,120 children, 47% of these children experienced adverse events.
For someone taking Keppra without a seizure disorder, these side effects serve no medical purpose whatsoever.
Common side effects include sleepiness, weakness, and dizziness. According to medical literature, these typically occur during the first 4 weeks of treatment and may impair the ability to drive or operate machinery safely.
Now, this is where it gets interesting. Research has shown that levetiracetam has been reported to induce psychiatric side effects in 13.3% of adults, with only 0.7% presenting with severe symptoms such as depression, agitation, or hostility. A case report published in Case Reports in Psychiatry documented levetiracetam-induced behavioral abnormalities including agitation, hostility, psychosis, and mood symptoms.
Behavioral and Psychiatric Effects
The behavioral side effects of Keppra deserve special attention. These aren’t minor inconveniences—they can fundamentally alter personality and behavior.
Reported behavioral changes include:
- Aggression and hostility
- Agitation and irritability
- Anxiety and depression
- Mood swings and emotional instability
- Suicidal thoughts or behavior
- Psychotic symptoms in severe cases
Discussions on medical forums frequently mention these psychiatric effects. Users describe personality changes that strain relationships and impact quality of life—all while managing epilepsy or during treatment. For someone without epilepsy taking Keppra unnecessarily, these effects would be purely detrimental.
What Happens to the Brain on Unnecessary Keppra
Levetiracetam is rapidly absorbed and has very high (96%) bioavailability. The medication crosses the blood-brain barrier and begins affecting neurotransmitter release within hours of administration.
For a brain not experiencing seizure activity, this intervention disrupts normal neural function. The medication is slowing down electrical signals that don’t need slowing, interfering with neurotransmitter systems that are already functioning properly.
Real talk: the brain evolved sophisticated mechanisms for regulating its own electrical activity. Artificially suppressing these mechanisms without medical cause can lead to unintended consequences.
The exact mechanisms of levetiracetam remain incompletely understood even by researchers. Taking a medication whose full neurological effects aren’t entirely mapped out—without having the condition it treats—represents a particularly risky gamble.
The Withdrawal Problem
Here’s something many people don’t consider: stopping Keppra isn’t straightforward, even for people who shouldn’t have been taking it in the first place.
Antiseizure medications typically require gradual tapering rather than abrupt discontinuation. Discussions on medical forums like Mayo Clinic Connect reveal that patients often struggle to find proper weaning protocols even under medical supervision.
One documented case involved a patient who had a single seizure in sleep, underwent multiple diagnostic tests (MRI, CT scan, EEG) that came back normal, and was determined through sleep study to have sleep apnea rather than epilepsy. Yet the weaning process from Keppra still required careful medical management.
For someone who never needed the medication, the withdrawal process adds another layer of unnecessary medical intervention and risk.
Specific Scenarios: When Someone Might Take Keppra Unnecessarily
Misdiagnosis Situations
Sometimes individuals are prescribed Keppra based on an initial diagnosis that later proves incorrect. One documented case involved a patient who had a single seizure in sleep, underwent multiple diagnostic tests (MRI, CT scan, EEG) that came back normal, and was determined through sleep study to have sleep apnea rather than epilepsy.
In such cases, the patient is taking Keppra unnecessarily—experiencing all the medication’s side effects while the actual underlying condition (sleep apnea) remains the true treatment target.
Prophylactic Use Without Indication
In some medical contexts, Keppra is used prophylactically—to prevent seizures that might occur due to brain injury, surgery, or other acute conditions. But when these conditions don’t materialize or the risk assessment was incorrect, patients may continue taking the medication longer than medically justified.
Medication Errors or Mix-ups
Pharmacy errors, though rare, can result in someone receiving Keppra when prescribed a different medication entirely. Taking even a few doses before catching the error exposes the individual to unnecessary risks.
Comparison: Generic vs. Brand-Name Keppra
Research published in Drug Design, Development and Therapy examined the safety of switching from brand-name to generic levetiracetam. Among 151 subjects who switched to generic formulations, increased frequency of seizures was noted in 9 patients (6%) during follow-up visits.
This finding matters because it demonstrates how even subtle formulation differences can impact outcomes in people who actually need the medication. For someone taking Keppra unnecessarily, brand versus generic becomes a moot point—neither formulation provides benefit when no seizure disorder exists.
| Aspect | Brand-Name Keppra | Generic Levetiracetam |
|---|---|---|
| Active Ingredient | Levetiracetam | Levetiracetam |
| Bioavailability | 96% | 96% (required by FDA) |
| Side Effect Profile | Identical risk spectrum | Identical risk spectrum |
| DRESS Risk | Present | Present |
| Behavioral Effects | 13.3% in adults | 13.3% in adults |
Medical Monitoring Required for Keppra
Patients legitimately taking Keppra require ongoing medical monitoring. This typically includes regular blood tests, neurological assessments, and evaluation for behavioral changes.
For someone taking the medication without medical necessity, this monitoring either doesn’t occur (increasing risk) or wastes medical resources that could serve patients with genuine needs.
Healthcare providers monitor for:
- Behavioral or mood changes requiring intervention
- Signs of DRESS syndrome or allergic reactions
- Coordination problems or excessive sedation
- Suicidal ideation or severe depression
- Drug interactions with other medications
Drug Interactions and Unnecessary Risk
Levetiracetam can interact with other medications, potentially altering their effectiveness or increasing side effect risks. Common interactions occur with other central nervous system depressants, certain psychiatric medications, and other antiseizure drugs.
Someone taking Keppra unnecessarily faces these interaction risks without any offsetting medical benefit. If they later require treatment for an unrelated condition, the presence of levetiracetam in their system may complicate treatment decisions.
What to Do If Taking Keppra Without a Seizure Disorder
Anyone currently taking Keppra who suspects they don’t have a seizure disorder should take these steps:
Do not stop the medication abruptly. Even unnecessary Keppra requires careful discontinuation to avoid withdrawal effects and potential rebound phenomena.
Schedule an appointment with the prescribing physician immediately. Bring documentation of any diagnostic tests that suggest the absence of a seizure disorder. Request a comprehensive reevaluation of the diagnosis and treatment plan.
Consider seeking a second opinion from a neurologist who specializes in epilepsy. Epilepsy specialists can provide expert assessment of whether the original diagnosis was accurate and whether continued treatment is medically indicated.
Document all side effects experienced while on the medication. This information helps healthcare providers assess the risk-benefit balance and make informed decisions about discontinuation.
If the decision is made to discontinue Keppra, work closely with medical professionals to develop an appropriate tapering schedule. The rate of dose reduction varies based on individual factors including duration of use, dosage level, and concurrent medications.
The Broader Context of Antiseizure Medication Use
Keppra represents just one example of a broader pattern in healthcare: the importance of precision diagnosis and appropriate prescribing.
Antiseizure medications as a class carry significant side effect burdens. They’re powerful drugs that alter fundamental brain chemistry. Using them requires clear medical justification—a diagnosed condition that warrants their specific mechanism of action.
Evidence from systematic reviews shows that even in appropriate use cases, balancing efficacy against adverse events remains challenging. An evidence map of levetiracetam for epilepsy published in Neuropsychiatric Disease and Treatment found that while the drug demonstrates clear efficacy for seizure control, safety considerations remain paramount in treatment decisions.
That said, when epilepsy exists, treatment is essential. The Epilepsy Foundation notes that for approximately 1 in 3 people with epilepsy, medications aren’t fully effective at controlling seizures. This underscores both the seriousness of the condition and the complexity of treatment.
But these treatment challenges apply only to people who actually have epilepsy. For everyone else, Keppra offers no benefits—only risks.
Frequently Asked Questions
Keppra itself doesn’t typically cause seizures in people without epilepsy, but abruptly stopping the medication after regular use can potentially trigger seizures even in individuals who never had a seizure disorder. This represents one of the paradoxical risks of taking antiseizure medication unnecessarily—it can create dependency that makes safe discontinuation challenging.
Most common side effects like dizziness, sleepiness, and weakness resolve within a few days to weeks after complete discontinuation. However, behavioral and mood changes may take longer to fully resolve, sometimes persisting for several weeks after the last dose. The timeline varies based on dosage, duration of use, and individual metabolism.
A single dose of Keppra is unlikely to cause serious harm in most individuals without epilepsy, though it may cause drowsiness, dizziness, or mild coordination problems. The more serious risks like DRESS syndrome typically require sustained exposure. That said, even single doses can trigger allergic reactions in susceptible individuals, and there’s no medical benefit to taking even one unnecessary dose.
While some clinicians have explored off-label uses of levetiracetam for conditions like migraine prevention or certain psychiatric disorders, these applications remain outside FDA-approved indications and lack robust supporting evidence. Off-label prescribing should only occur when a qualified specialist determines potential benefits outweigh risks for a specific patient’s situation.
Early warning signs include unusual tiredness or weakness, personality changes or increased irritability, coordination problems or unsteadiness, and mood changes like increased anxiety or sadness. More serious warning signs requiring immediate medical attention include severe rash, fever with swollen glands, extreme mood changes or thoughts of self-harm, and difficulty breathing or swelling of the face or throat.
Diagnosis of epilepsy typically involves multiple components: detailed medical history including description of seizure-like events, electroencephalogram (EEG) to detect abnormal brain electrical activity, brain imaging (MRI or CT) to identify structural abnormalities, and sometimes prolonged video-EEG monitoring to capture and characterize seizure events. Keppra should only be prescribed when these diagnostic tools confirm a seizure disorder that warrants treatment.
Some patients with epilepsy report decreased effectiveness of levetiracetam over time, though true pharmacological tolerance is not well-established in the literature. More commonly, changes in seizure control relate to disease progression, medication adherence issues, or development of drug interactions. For someone without epilepsy, the concept of tolerance is irrelevant since there’s no therapeutic effect to diminish.
Conclusion: The Clear Medical Consensus
The medical consensus remains unambiguous: taking Keppra without a diagnosed seizure disorder represents an unnecessary medical risk without any corresponding benefit.
The FDA’s warning about DRESS syndrome demonstrates that even rare serious reactions carry real consequences. The well-documented behavioral and psychiatric effects—affecting more than 13% of adult users—represent a substantial burden when endured without medical justification.
Look, medications exist to treat specific conditions. When the condition doesn’t exist, the medication becomes purely problematic.
If there’s any question about whether Keppra is medically necessary in a specific situation, that question deserves thorough investigation by qualified healthcare providers. Diagnostic uncertainty should prompt additional evaluation, not continued exposure to a powerful medication with known serious risks.
For anyone currently taking Keppra and questioning whether it’s medically necessary: seek medical consultation immediately. But never discontinue the medication without professional guidance, as even unnecessary Keppra requires careful management during discontinuation.
The bottom line? Keppra saves lives and improves quality of life for people with seizure disorders. For everyone else, it’s a medication best avoided entirely.
