Quick Summary: Using expired tirzepatide carries significant risks including reduced potency, unpredictable effectiveness, and potential safety concerns. While the medication may not become immediately toxic after expiration, its therapeutic benefits decline over time, making blood sugar control and weight loss unreliable. Proper refrigerated storage and adherence to expiration dates are essential for maintaining medication effectiveness.
Every medication comes with an expiration date for a reason. For tirzepatide users—whether managing type 2 diabetes or pursuing weight loss goals—that date represents more than just a formality.
It’s the manufacturer’s guarantee that the medication will work as intended.
But what actually happens when tirzepatide passes its expiration date? Does it become dangerous overnight, or does it simply lose effectiveness gradually? And what about compounded tirzepatide, which behaves differently from commercial brands like Mounjaro or Zepbound?
Understanding these distinctions isn’t just academic. According to FDA Adverse Event Reporting System data, incorrect dose administration has become a significant concern with tirzepatide, with reports increasing from 1,248 in 2022 to 9,800 in 2024. While not all these incidents involve expired medication, they highlight how critical proper medication handling has become.
Here’s what you need to know about expired tirzepatide, storage requirements, and the real risks involved.
Understanding Tirzepatide Expiration Dates
Tirzepatide is a dual glucose-dependent insulinotropic polypeptide (GIP) and glucagon-like peptide-1 (GLP-1) receptor agonist. That complex mechanism means the medication contains delicate peptide structures that degrade over time.
The expiration date stamped on your tirzepatide vial or pen isn’t arbitrary. It represents the point at which the manufacturer can no longer guarantee the medication maintains its full potency under proper storage conditions.
Commercial tirzepatide products like Mounjaro and Zepbound undergo rigorous stability testing. These tests determine how long the active ingredient remains stable at specified temperatures.
Commercial vs. Compounded Tirzepatide
The shelf life question becomes more complicated with compounded tirzepatide. Unlike FDA-approved commercial products, compounded medications come from specialized pharmacies that mix ingredients on-site.
Compounded tirzepatide behaves very differently from standardized commercial medications. The shelf life depends on the compounding pharmacy’s specific formulation, sterility procedures, and storage recommendations.
Real-world safety data remains limited for compounded versions. Without the extensive stability testing required for FDA-approved drugs, determining exact potency over time becomes challenging.
Does Expired Tirzepatide Still Work?
The short answer? Maybe, but unreliably.
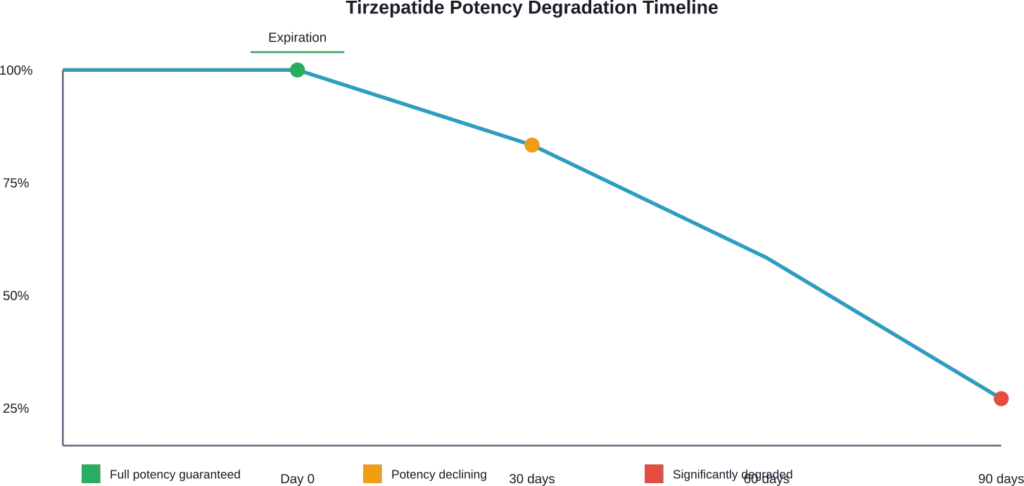
Medications don’t typically transform into toxic substances the day after expiration. Instead, they undergo gradual chemical degradation that reduces potency.
For tirzepatide, this degradation means the peptide chains that activate GIP and GLP-1 receptors break down. The medication remains physically present in the vial, but its therapeutic effect diminishes.
Here’s the problem: there’s no reliable way to confirm remaining potency without laboratory analysis. You can’t look at the solution, smell it, or test it at home to determine if it still contains the full dose.
Clinical trials for tirzepatide demonstrated mean weight reductions, with some analyses showing reductions ranging from 15% to 22% of initial body weight over extended periods. But those results depend on consistent, full-potency dosing.
Using degraded medication means you’re essentially taking a reduced dose without knowing how much reduction has occurred. That unpredictability undermines treatment goals.
Safety Risks of Using Expired Tirzepatide
While expired tirzepatide likely won’t cause acute toxicity, several safety concerns emerge.
Reduced Therapeutic Effect
The primary risk is treatment failure. For type 2 diabetes management, reduced potency means inadequate blood sugar control. For weight loss, it means slower or stalled progress.
According to pharmacovigilance data from FAERS analysis covering 2022 through 2025, gastrointestinal adverse events represent the most common issues with tirzepatide, with nausea having a reporting odds ratio (ROR) of 4.01 (95% CI 3.85–4.19). If you’re experiencing side effects but not therapeutic benefits due to degraded medication, you’re getting the worst of both outcomes.
Unpredictable Dosing
Degradation doesn’t happen uniformly. Temperature fluctuations, light exposure, and time all affect how quickly potency declines.
This creates a dosing nightmare. One expired vial might retain 80% potency while another from the same batch might have dropped to 60%. There’s no way to know without laboratory testing.
Contamination Concerns
Once a medication passes its expiration date, the preservatives and stabilizers that prevent bacterial growth may also lose effectiveness. This becomes particularly concerning for compounded tirzepatide stored in multi-dose vials.
Every time you insert a needle into a vial, you introduce potential contamination. Fresh medication with active preservatives handles this risk. Expired medication with degraded preservatives? That’s a different story.
| Risk Factor | Potential Consequence | Severity |
|---|---|---|
| Reduced potency | Inadequate blood sugar control or weight loss | Moderate to High |
| Unpredictable dosing | Inconsistent results, difficulty adjusting treatment | Moderate |
| Degraded preservatives | Increased contamination risk in multi-dose vials | Moderate |
| Chemical breakdown products | Unknown effects from degraded peptides | Low to Moderate |
| False sense of treatment | Delayed proper medical care | High |
Proper Storage Requirements for Tirzepatide
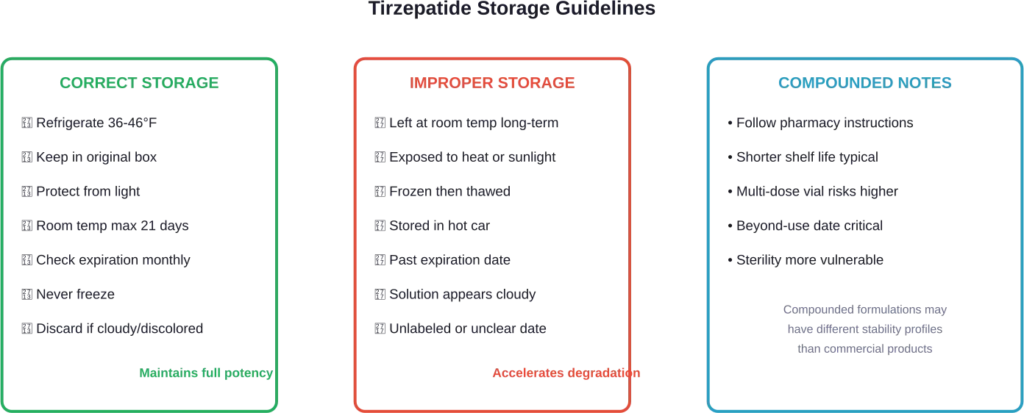
Preventing premature degradation starts with proper storage. Commercial tirzepatide products require refrigeration between 36°F and 46°F (2°C and 8°C).
Once removed from refrigeration, tirzepatide pens can remain at room temperature below 86°F (30°C) for up to 21 days. After that window, the medication should be discarded even if the printed expiration date hasn’t arrived.
The Storage Mistakes That Destroy Potency
Temperature fluctuations cause the most damage. Leaving tirzepatide in a hot car, storing it near a heat source, or forgetting it on the counter overnight all accelerate degradation.
Freezing presents another problem. If tirzepatide freezes, it should be discarded immediately. Freezing irreversibly damages the peptide structure, rendering the medication ineffective even after thawing.
Light exposure also matters. Tirzepatide should remain in its original packaging until use to protect it from light degradation.
Compounded Tirzepatide Storage Considerations
Compounded tirzepatide often comes with more stringent storage requirements and shorter beyond-use dates. Some compounding pharmacies assign 30-60 day beyond-use dates regardless of the theoretical stability of the active ingredient.
This conservative approach reflects the reality that compounded medications lack the extensive stability data backing commercial products. Without that data, pharmacies err on the side of caution.
What to Do If Your Tirzepatide Has Expired
Found an expired vial in your refrigerator? Here’s what to do.
First, don’t use it. The risk-benefit calculation doesn’t favor using expired tirzepatide. The potential for reduced effectiveness outweighs any cost savings from trying to extend the medication’s life.
Second, dispose of it properly. Don’t flush medications down the toilet or throw them in household trash. Many pharmacies offer medication take-back programs. The FDA also provides guidance on safe medication disposal.
Third, contact your healthcare provider for a new prescription. If cost concerns drove you to consider using expired medication, discuss this openly. Alternative solutions might include:
- Patient assistance programs from manufacturers
- Generic alternatives as they become available
- Adjusting treatment plans based on budget constraints
- Exploring compounding pharmacy options if appropriate
Real talk: the healthcare system makes medication access challenging. But using expired medication isn’t the solution. It compromises treatment outcomes while still exposing you to potential side effects.
Recognizing Degraded Tirzepatide
Beyond checking expiration dates, you should inspect tirzepatide before each use.
Fresh tirzepatide appears as a clear, colorless to slightly yellow solution. If the solution looks cloudy, contains particles, or has changed color significantly, don’t use it regardless of the expiration date.
For compounded tirzepatide with B12, the solution may have a slight pink tint from the B12. But it should still appear clear, not cloudy or murky.
Any visible changes suggest degradation or contamination. Trust your eyes—when in doubt, throw it out.
The Broader Context: GLP-1 Medication Safety
Tirzepatide belongs to the GLP-1 receptor agonist class, which also includes semaglutide. These medications have transformed type 2 diabetes and obesity treatment, but their rapid adoption has introduced safety monitoring challenges.
Post-marketing surveillance data reveals important safety signals. Among 65,974 adverse event reports for tirzepatide analyzed from FAERS data (2022–2025), the majority originated from the U.S. (96%), with middle-aged females (40–59 years; 67%) most frequently affected. Incorrect dose administration topped the adverse event list, highlighting how critical proper medication handling has become.
The FDA has issued warning letters to compounding pharmacies for inadequate quality control procedures. A 2025 FDA warning letter cited a compounding facility for lacking proper inspection protocols in their visual inspection program, with no mention of 100% inspection, Acceptance Quality Limit (AQL) inspection, operator qualification, or classification of defects.
These regulatory actions underscore the importance of obtaining tirzepatide from reputable sources and following storage and expiration guidelines precisely.
Cost Considerations and Medication Access
Let’s address the elephant in the room: tirzepatide costs a lot. That financial burden tempts people to stretch medications beyond expiration dates.
But consider what you’re actually doing. You’re paying for medication that may not work at full strength. You’re accepting the side effects—nausea, vomiting, diarrhea—without the guarantee of therapeutic benefit. And you’re potentially delaying effective treatment while your health condition progresses.
From a pure cost-effectiveness standpoint, using expired medication is a losing proposition. You might save money upfront, but you’re sacrificing the very outcome you’re paying to achieve.
Frequently Asked Questions
There’s no definitive answer because degradation rates vary based on storage conditions. While tirzepatide doesn’t become immediately dangerous after expiration, potency declines progressively. Some degradation begins even before the expiration date if storage conditions aren’t ideal. The expiration date represents the last day the manufacturer guarantees full potency—using it beyond that point means accepting unpredictable effectiveness.
Obvious signs of degradation include cloudiness, discoloration, or visible particles in the solution. However, potency loss can occur without any visible changes. Tirzepatide might look perfectly normal but have significantly reduced effectiveness. Visual inspection catches contamination or severe degradation but won’t confirm potency levels.
Commercial products like Mounjaro and Zepbound carry expiration dates based on extensive stability testing under FDA requirements. Compounded tirzepatide typically has shorter beyond-use dates assigned by the compounding pharmacy, often 30-60 days, because it lacks the same stability data. Compounded versions generally have more conservative dating due to variables in compounding processes and formulations.
Expired tirzepatide is unlikely to cause acute illness or toxicity in most cases. The primary concern is reduced effectiveness rather than new harmful effects. However, degraded preservatives in multi-dose vials could potentially allow bacterial contamination, which poses infection risk. The greater danger is continuing a treatment plan with medication that no longer delivers therapeutic benefits.
Yes, freezing causes irreversible damage to tirzepatide’s peptide structure. Even if the medication thaws and appears normal, the freezing process disrupts the molecular configuration necessary for effectiveness. Any tirzepatide that has been frozen should be discarded immediately, regardless of its expiration date or how recently it was filled.
Don’t flush tirzepatide or throw it in regular trash. Many pharmacies offer medication take-back programs where you can drop off expired medications for proper disposal. The FDA recommends mixing unwanted medications with undesirable substances like coffee grounds in a sealed bag before disposal if take-back options aren’t available. Check FDA guidance or ask your pharmacist for local disposal options.
Absolutely. Temperature excursions, light exposure, and freezing all accelerate degradation. Tirzepatide left at room temperature beyond the recommended 21-day window loses potency faster than properly refrigerated medication. Heat exposure is particularly damaging—even a few hours in a hot car can compromise effectiveness. The expiration date assumes proper storage throughout the medication’s life; improper storage invalidates that timeline.
Conclusion: Medication Safety Isn’t Negotiable
Using expired tirzepatide represents an unacceptable gamble with treatment outcomes and potentially with safety.
The medication loses potency unpredictably after expiration. You can’t determine remaining effectiveness through visual inspection or home testing. And the therapeutic goals you’re trying to achieve—whether blood sugar control or weight loss—depend on consistent, full-strength dosing.
Proper storage, adherence to expiration dates, and regular prescription refills aren’t optional extras. They’re fundamental requirements for effective treatment.
If cost concerns make medication access difficult, have that conversation with your healthcare provider. Solutions exist that don’t involve compromising medication quality or effectiveness. Patient assistance programs, alternative therapies, and adjusted treatment plans can address financial constraints while maintaining treatment integrity.
Your health deserves medication that works. Don’t settle for expired products that might work. Check your tirzepatide today, verify proper storage, and confirm you’re within the expiration window. If not, it’s time for a refill.
