Quick Summary: Splitting an atom—nuclear fission—releases massive energy as the atom’s nucleus breaks into smaller fragments, emitting neutrons and radiation. This process powers nuclear reactors and atomic weapons, converting tiny amounts of mass into enormous heat through Einstein’s E=mc². The energy release occurs because the fragments are more stable than the original atom, and the mass difference becomes pure energy.
The word “atom” comes from Greek for indivisible. But here’s the thing—atoms are anything but permanent.
When you split an atom, particularly a heavy one like uranium-235, you trigger one of nature’s most powerful energy-releasing processes. The nucleus breaks apart, fragments fly off at incredible speeds, and neutrons scatter in every direction. The energy released from a single split atom could power a light bulb for a fraction of a second. Scale that up to trillions of atoms, and you’ve got a nuclear reactor—or a weapon.
So what actually happens during this split? And why does something so small create such enormous energy?
The Basics of Atomic Structure
Every atom consists of a nucleus surrounded by electrons. The nucleus contains protons and neutrons packed tightly together. For most atoms, this arrangement remains stable indefinitely.
According to the U.S. Department of Energy, the nucleus represents nearly all the atom’s mass despite occupying a tiny fraction of its volume. Protons carry a positive charge, neutrons carry no charge, and electrons orbit with a negative charge.
In lighter elements like carbon or oxygen, the nucleus stays bound together easily. But in heavy elements like uranium-235, which contains 92 protons and 143 neutrons, the nucleus becomes less stable. The arrangement of these 235 particles creates conditions ripe for splitting.
Why Heavy Atoms Are Easier to Split
Heavy atomic nuclei are somewhat unstable. The electromagnetic force pushing protons apart begins to compete with the strong nuclear force holding everything together.
Uranium-235 sits on this edge of instability. Add just one more neutron—an outside nudge—and the whole structure can disintegrate. That’s the foundation of nuclear fission.
The Fission Process Explained
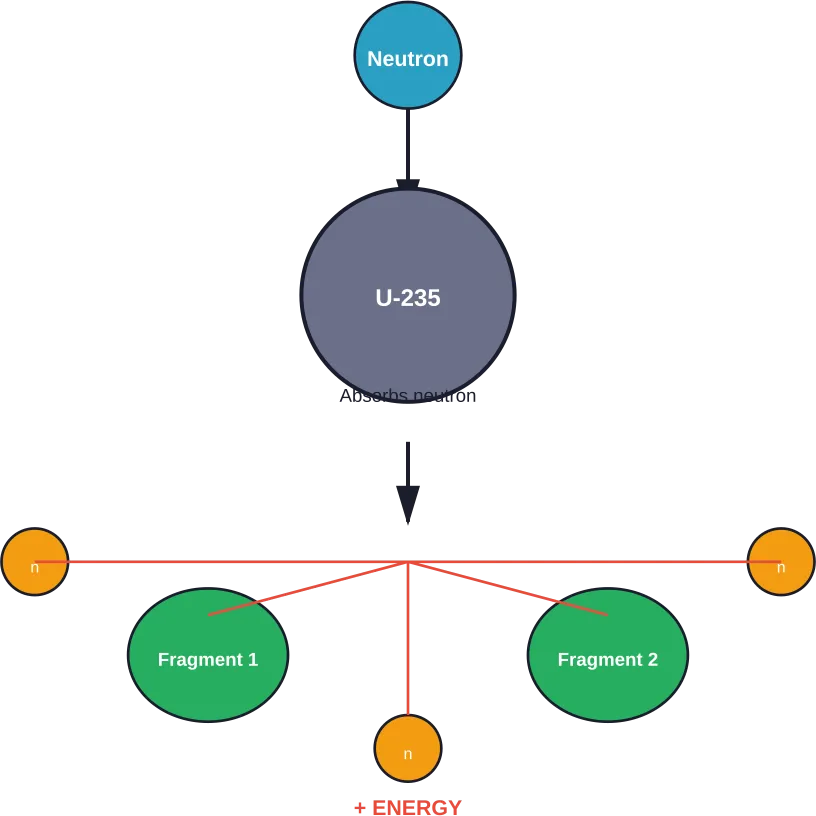
Nuclear fission occurs when a neutron strikes an atom’s nucleus, causing it to split into smaller fragments. According to the U.S. Nuclear Regulatory Commission, this creates fragments of the original nucleus plus new neutrons that can trigger additional reactions.
Here’s what happens step by step when a uranium-235 atom splits:
- A neutron approaches the U-235 nucleus and gets absorbed
- The nucleus briefly becomes uranium-236, an excited and unstable state
- Within a fraction of a second, the nucleus splits into two smaller nuclei
- The split releases two or three additional neutrons
- Massive amounts of energy release as heat and radiation
The MIT Nuclear Reactor Laboratory notes that each time a U-235 nucleus splits, it releases two or three neutrons. These neutrons can then strike other uranium atoms, creating a chain reaction.
The Chain Reaction Phenomenon
When one split atom releases neutrons that strike other atoms, causing them to split and release more neutrons, a chain reaction begins. This self-sustaining process can continue as long as fissile material remains available.
In nuclear reactors, control rods absorb excess neutrons to maintain a steady, controlled reaction. In atomic weapons, the reaction proceeds uncontrolled, with billions of atoms splitting within microseconds.
Where Does the Energy Come From?
The energy released during fission comes from Einstein’s famous equation: E=mc². A tiny amount of mass converts directly into energy.
When a uranium-235 nucleus splits, the combined mass of the fragments, neutrons, and radiation is slightly less than the original atom. That missing mass—typically less than one percent—becomes pure energy.
According to Stanford University’s Understand Energy Learning Hub, nuclear fission produces enormous amounts of energy because atomic bonds are millions of times stronger than chemical bonds. Burning coal releases energy by rearranging electrons between atoms. Fission releases energy by breaking apart the nucleus itself.
The Department of Energy notes that nuclear energy accounts for 9% of the world’s electricity generation. This demonstrates how efficiently mass converts to energy at the atomic level.
Quantifying the Energy Release
A single uranium-235 fission event releases approximately 200 million electron volts of energy. That might sound small, but it’s about 50 million times more energy than burning a single carbon atom in coal.
Scale that to a kilogram of uranium-235, and complete fission would release energy equivalent to burning roughly 3 million kilograms of coal. That’s the power of splitting atoms.
What Happens to the Fragments?
The fragments created during fission aren’t identical. When uranium-235 splits, it typically produces two unequal fragments—one larger, one smaller. Common fragment pairs include barium and krypton, or strontium and xenon.
These fragments are themselves radioactive. They contain excess neutrons and need to shed energy to reach stability. This process produces radiation over time, creating what we know as radioactive waste.
According to the International Atomic Energy Agency, radioactive waste makes up a small portion of all waste but requires careful management due to its long-term radiation emissions. The fragments decay over periods ranging from seconds to thousands of years.
| Fission Product | Half-Life | Radiation Type |
|---|---|---|
| Iodine-131 | 8 days | Beta, Gamma |
| Strontium-90 | 29 years | Beta |
| Cesium-137 | 30 years | Beta, Gamma |
| Plutonium-239 | 24,000 years | Alpha |
Radiation Emissions
The fragments and neutrons released during fission produce three types of radiation: alpha particles, beta particles, and gamma rays. Alpha particles consist of two protons and two neutrons. Beta particles are high-speed electrons. Gamma rays are electromagnetic radiation.
This radiation makes spent nuclear fuel dangerous. But it also makes nuclear medicine possible, enabling millions of medical procedures annually according to the IAEA.
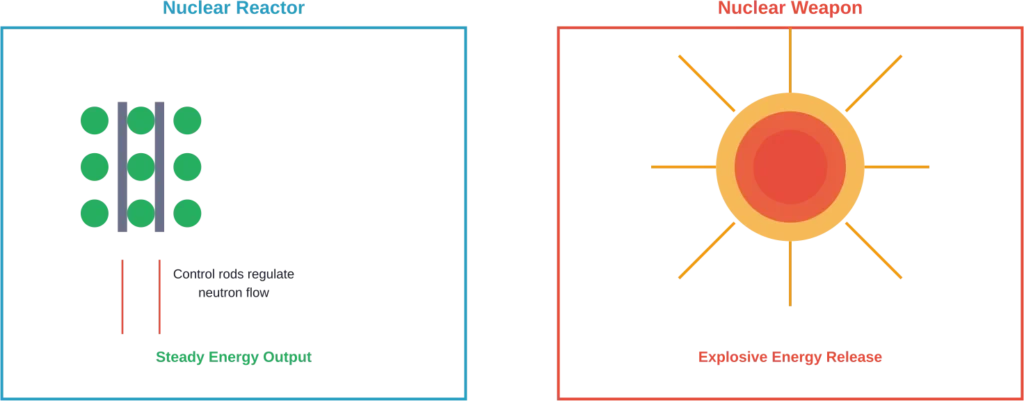
Controlled vs. Uncontrolled Fission
The difference between a nuclear reactor and a nuclear weapon comes down to control. Both use the same fundamental process—splitting atoms—but manage the chain reaction differently.
Nuclear Reactors
In nuclear power plants, operators maintain a steady, controlled chain reaction. According to the Department of Energy, control rods made of materials that absorb neutrons slide between fuel rods to regulate the reaction rate.
When operators want more power, they withdraw control rods slightly, allowing more neutrons to trigger fission events. To reduce power, they insert the rods further, absorbing more neutrons and slowing the reaction.
The heat generated from controlled fission boils water, creating steam that spins turbines to generate electricity. Pressurized water reactors and boiling water reactors both use this principle, with slight variations in design.
Nuclear Weapons
Nuclear weapons allow an uncontrolled chain reaction to proceed as rapidly as possible. In a fraction of a second, a critical mass of uranium-235 or plutonium-239 undergoes trillions of fission events simultaneously.
The energy releases so rapidly that it creates an explosion. The heat generates temperatures hotter than the sun’s surface, creating the characteristic mushroom cloud and devastating blast wave.
Can You Split Any Atom?
Technically, yes—but practically, only certain atoms undergo fission easily. Heavy atoms with large, unstable nuclei split most readily. Uranium-235, uranium-233, and plutonium-239 are the primary fissile materials used in reactors and weapons.
Lighter atoms can theoretically split, but they require enormous energy input. The energy needed to split a light atom like carbon or oxygen exceeds the energy the split would release. That makes fission energetically unfavorable for most elements.
Community discussions on nuclear physics forums note that splitting a “normal” atom from everyday matter wouldn’t produce an explosion. The atom would simply break apart and likely recombine or form different molecules. The key to energy-producing fission is splitting atoms that release more energy than it takes to split them.
Spontaneous Fission
Some heavy atoms split without any external trigger. Uranium-238, for example, occasionally undergoes spontaneous fission. The process occurs rarely—most U-238 atoms decay through alpha emission instead—but it happens.
The IAEA notes that spontaneous fission occurs naturally in heavy elements but at rates too slow for energy production. Induced fission, where neutrons trigger the split, provides the controllable reaction needed for practical applications.
The Discovery of Atom Splitting
Nuclear fission was discovered in December 1938 by Otto Hahn and Fritz Strassmann. Physicist Lise Meitner and her nephew Otto Frisch provided the theoretical explanation for what was happening.
According to the IAEA’s historical documentation, Meitner and Frisch first described the phenomenon in a 1939 paper. They explained that the nucleus absorbed a neutron, became unstable, and split into smaller parts—a process they named “fission” after biological cell division.
This discovery marked a pivotal moment in nuclear science. Within years, the first controlled nuclear chain reaction occurred at the University of Chicago in December 1942. Nuclear technology development accelerated rapidly from there.
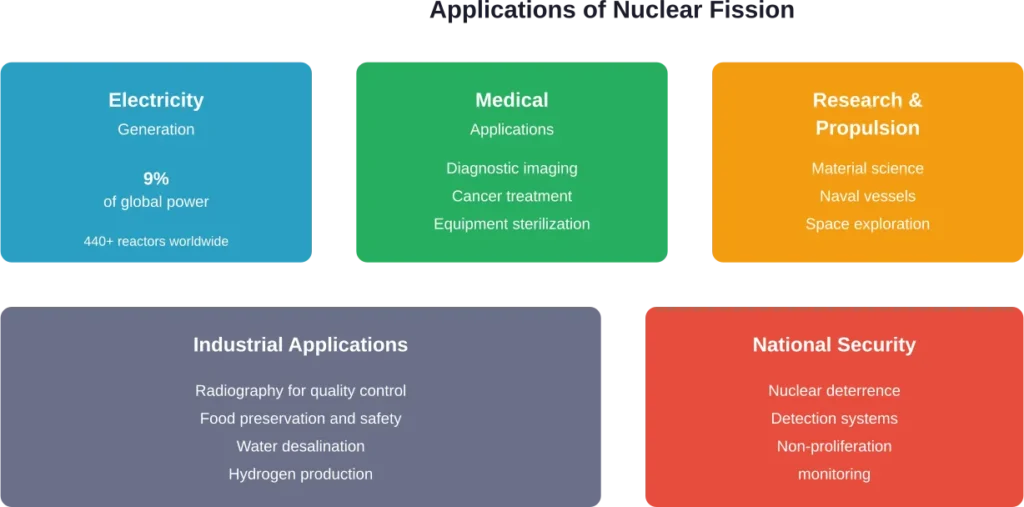
Modern Applications of Atom Splitting
Nuclear fission powers approximately 9% of global electricity generation according to the IAEA. Countries like France generate over 70% of their electricity from nuclear power, demonstrating the technology’s scalability.
Beyond electricity generation, fission enables medical applications. Radioactive isotopes produced in research reactors help diagnose and treat cancer, sterilize medical equipment, and conduct millions of procedures annually.
Nuclear Medicine
Medical isotopes produced through fission and neutron activation include technetium-99m for diagnostic imaging, iodine-131 for thyroid treatment, and cobalt-60 for cancer radiotherapy. The IAEA reports these applications save countless lives.
Research and Propulsion
Research reactors enable neutron scattering experiments that reveal material structures at atomic scales. Nuclear propulsion powers submarines and aircraft carriers, providing vessels that can operate for decades without refueling.
The Environmental Impact
Nuclear fission produces zero carbon emissions during operation. According to the IAEA, fusion and fission both rank among the most environmentally friendly energy sources when measured by greenhouse gas emissions.
But nuclear power creates radioactive waste. Spent fuel contains fission products that remain radioactive for thousands of years. Managing this waste requires secure storage facilities designed to isolate materials from the environment.
According to Stanford University’s Understand Energy Learning Hub, the highly radioactive byproducts of fission energy must be secured away from people for hundreds of thousands of years, but there are no proven long-term solutions for storage.
Safety Considerations
Nuclear accidents at Three Mile Island, Chernobyl, and Fukushima demonstrated that when fission reactions lose control, consequences can be severe. Modern reactor designs incorporate passive safety features that shut down reactions automatically without human intervention.
Statistically, nuclear energy causes fewer deaths per unit of electricity generated than fossil fuels. But the potential for catastrophic accidents and long-lived radioactive contamination makes nuclear safety paramount.
Fission vs. Fusion
Nuclear fission splits heavy atoms apart. Nuclear fusion combines light atoms together. Both processes release energy, but they work through opposite mechanisms.
According to the U.S. Nuclear Regulatory Commission, fusion occurs when two light nuclei combine to form a heavier nucleus. This process powers the sun, where hydrogen atoms fuse into helium under extreme temperature and pressure.
Fusion produces no long-lived radioactive waste and uses abundant fuel sources like hydrogen. However, achieving the temperatures and pressures needed for controlled fusion on Earth remains technically challenging. As of 2026, no fusion reactor has achieved sustained net energy production for commercial electricity generation.
| Characteristic | Nuclear Fission | Nuclear Fusion |
|---|---|---|
| Process | Splits heavy atoms | Combines light atoms |
| Fuel | Uranium-235, Plutonium-239 | Hydrogen isotopes |
| Waste | Long-lived radioactive waste | Minimal radioactive waste |
| Temperature | A few hundred degrees C | Millions of degrees C |
| Current Status | Operational worldwide | Experimental only |
| Energy Output | High | Potentially higher |
Recent Scientific Advances
Researchers continue refining our understanding of exactly what happens during atom splitting. Theoretical physicists have created the first fully microscopic characterization of the fission moment, revealing new insights into how the nucleus deforms before splitting.
These simulations help explain why certain isotopes split more readily than others and how fragment distributions occur. The research improves predictions for reactor fuel behavior and waste composition.
Small modular reactors represent another advancement. These compact designs promise safer, more flexible nuclear power deployment. Several advanced reactor designs are in development.
Splitting Atoms in Your Body
Community discussions occasionally ask what would happen if an atom in a human body split. The answer? Almost nothing noticeable.
Your body contains mostly light elements—carbon, hydrogen, oxygen, nitrogen—that don’t undergo fission. Even if a single heavy atom like potassium-40 (which is naturally radioactive) split, the energy would dissipate as heat across nearby molecules.
Your body receives more radiation from natural background sources—cosmic rays, radon gas, radioactive elements in soil—than from occasional atomic events inside cells. Radiation becomes dangerous at high doses, not from individual atomic splits.
The Future of Atom Splitting
Nuclear fission will likely remain part of the global energy mix for decades. The International Atomic Energy Agency projects significant growth in nuclear capacity, particularly in Asia and the Middle East. China is expected to surpass the U.S. in nuclear power generation capacity by 2030.
Advanced reactor designs promise improved safety, reduced waste, and higher efficiency. Thorium reactors, fast-neutron reactors, and molten salt reactors all aim to address limitations of current technology.
But challenges remain. Public perception, waste management, construction costs, and nuclear weapons proliferation concerns all complicate nuclear energy expansion.
Frequently Asked Questions
No. Splitting a single atom releases energy, but not an explosion. Explosions occur only when billions of atoms split in an uncontrolled chain reaction within microseconds, as in nuclear weapons. Nuclear reactors split atoms continuously in a controlled manner without exploding, using the heat for electricity generation.
Not realistically or safely. Splitting atoms requires either highly enriched fissile material (uranium-235 or plutonium-239) and specialized equipment, or particle accelerators that create the conditions for fission. These materials and tools are heavily regulated, dangerous, and inaccessible to the general public. Natural radioactive decay in household smoke detectors involves atomic transformation but not fission.
A single uranium-235 fission event releases approximately 200 million electron volts of energy. While this sounds large, it equals only about 3.2 × 10⁻¹¹ joules—barely enough to lift a grain of sand one millimeter. The enormous energy from nuclear reactions comes from splitting trillions of atoms simultaneously.
Splitting an atom (fission) breaks the nucleus into two large fragments plus neutrons. Radioactive decay typically involves an atom emitting a small particle—an alpha particle, beta particle, or gamma ray—while the nucleus remains mostly intact. Fission is induced or spontaneous splitting; decay is gradual particle emission. Both processes release energy and create different elements.
We already use nuclear fission for about 9% of global electricity according to the IAEA. Expanding nuclear power faces challenges including high construction costs, radioactive waste management, safety concerns after accidents like Fukushima, and political opposition. Additionally, uranium resources are finite, though abundant enough for centuries at current usage rates. Nuclear energy is part of the solution but not a complete answer alone.
No. Fission requires uranium or plutonium fuel, which must be mined and processed. Once split, these atoms become different elements and can’t be reused as fission fuel. Uranium supplies are finite, making fission non-renewable. However, breeder reactors can convert non-fissile uranium-238 into fissile plutonium-239, extending fuel supplies significantly.
The two or three neutrons released when an atom splits can take several paths: they might strike other fissile atoms and trigger additional fission (continuing the chain reaction), get absorbed by non-fissile materials, escape the reactor core, or get absorbed by control rods. In reactors, operators manage neutron populations carefully to maintain steady energy output.
Conclusion
Splitting an atom unleashes one of nature’s most powerful energy-releasing processes. When a heavy nucleus like uranium-235 absorbs a neutron, it becomes unstable and breaks into smaller fragments, releasing additional neutrons and massive amounts of energy as heat and radiation.
This process—nuclear fission—powers reactors generating 9% of global electricity. It enables medical treatments that save lives. And in uncontrolled form, it creates devastating weapons.
The energy comes from Einstein’s E=mc², where a tiny amount of mass converts into enormous energy. The fragments are more stable than the original atom, and the mass difference becomes the energy we harness.
Understanding fission helps explain how nuclear power works, why radioactive waste persists for millennia, and why both tremendous benefits and serious risks accompany this technology. As humanity seeks carbon-free energy sources, the atom-splitting discovered in 1938 remains relevant nearly 90 years later.
Want to dive deeper into nuclear energy? Explore how modern reactor designs improve safety, how radioactive waste gets managed, and how fusion energy might complement or replace fission in the coming decades.
