Quick Summary: Stopping Ozempic typically leads to weight regain and rising blood sugar levels within weeks to months. Research shows participants regain approximately two-thirds of their prior weight loss within one year of discontinuation. Most people experience increased appetite, fluctuating blood sugar, and potential return of pre-treatment health markers.
Ozempic has transformed diabetes management and weight loss since its FDA approval in 2017. But what happens when the medication stops?
The short answer? Most changes reverse.
Research indicates that discontinuing semaglutide (the active ingredient in Ozempic and Wegovy) triggers significant physiological shifts. Understanding these changes helps set realistic expectations whether stopping by choice, due to side effects, or insurance coverage issues.
How Ozempic Works in Your Body
Semaglutide belongs to a class of medications called glucagon-like peptide-1 receptor agonists (GLP-1 RAs). These drugs mimic a natural hormone that regulates appetite and blood sugar.
The medication slows stomach emptying, reduces hunger signals to the brain, and stimulates insulin production when blood sugar rises. These combined effects create the appetite suppression and weight loss that made Ozempic famous beyond its original diabetes indication.
Once treatment stops, these mechanisms gradually shut down. The body returns to its pre-treatment state, though the timeline varies between individuals.
Why People Stop Taking Ozempic
According to medical experts, three main reasons drive discontinuation:
Side effects. Approximately 15 percent of patients experience significant adverse reactions. Gastrointestinal complaints are particularly common—research published in the Journal of Medical Internet Research found that 62% of reviewers reported gastrointestinal issues, though these didn’t significantly influence satisfaction ratings.
The FDA has been evaluating reports of suicidal thoughts or actions in patients treated with GLP-1 RAs, though preliminary evaluation does not suggest a causal link. Still, patients should tell their health care professional if they experience new or worsening depression, suicidal thoughts, or any unusual changes in mood or behavior.
Reaching goal weight. For those using Ozempic off-label for weight loss, insurance coverage often ends once target weight is achieved. The medication can be costly, making long-term use financially impossible for many without insurance coverage.
Supply issues or access barriers. Widespread demand has created periodic shortages. Additionally, inequities in access mean some patients can’t maintain consistent treatment.
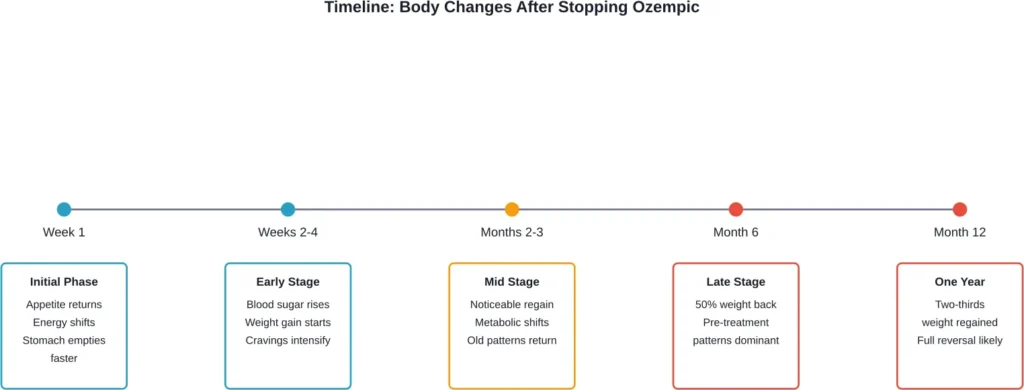
What Happens Immediately After Stopping Ozempic
The first few weeks reveal the most noticeable changes.
Appetite Returns with Vengeance
Two-thirds of patients reported reduced appetite, food cravings, or body weight while taking semaglutide. When treatment stops, hunger signals return—often stronger than before.
This rebound effect happens because the medication suppressed natural appetite regulation. The body compensates by ramping up hunger hormones once the drug clears the system.
Blood Sugar Fluctuations
For patients with type 2 diabetes, blood sugar levels typically begin rising within days to weeks. The medication’s glucose-regulating effects wear off as semaglutide leaves the body.
Monitoring becomes crucial during this transition. Some patients need alternative diabetes medications to prevent dangerous spikes.
Energy Level Changes
Many patients report consistent energy levels while on semaglutide. After stopping, energy fluctuations often mirror blood sugar instability and dietary changes.
Long-Term Effects of Stopping Ozempic
The months following discontinuation reveal the full scope of changes.
Weight Regain—The Two-Thirds Rule
Research published in medical journals confirms what patients fear most: weight returns rapidly.
One year after withdrawal of once-weekly subcutaneous semaglutide 2.4 mg and lifestyle intervention, participants regained two-thirds of their prior weight loss. Similar changes occurred in cardiometabolic variables.
This isn’t failure—it’s biology. The findings confirm the chronicity of obesity and suggest ongoing treatment is required to maintain weight reduction.
A case study reported in medical literature described a 35-year-old male who lost 22.7 kg over 120 days on oral semaglutide. Discontinuation led to significant regain, highlighting the challenge of maintaining results without continued therapy.
Metabolic Changes Reverse
Benefits beyond weight loss also diminish. Improvements in cardiovascular risk factors, inflammation markers, and insulin resistance typically revert toward baseline levels.
For patients who achieved better cardiac hemodynamics and metabolic benefits while on treatment, losing these gains represents a significant health setback.
Muscle Mass Considerations
Recent research indicates semaglutide impacts skeletal muscle to a similar extent as caloric restriction alone. While the medication reduces adipose tissue, there’s also loss of lean mass including skeletal muscle.
After stopping, weight regain often includes proportionally more fat than muscle compared to what was lost, potentially worsening body composition compared to pre-treatment status.
| Effect Category | While Taking Ozempic | After Stopping Ozempic |
|---|---|---|
| Appetite Control | Significantly suppressed | Returns to baseline or higher |
| Blood Sugar Levels | Stabilized and controlled | Rises, often requiring intervention |
| Weight Status | Progressive loss | Two-thirds regained within 12 months |
| Energy Levels | Generally consistent | Fluctuates with blood sugar changes |
| Cardiovascular Markers | Improved risk profile | Returns toward pre-treatment levels |
| Inflammation | Reduced systemic inflammation | Increases as weight returns |
Discontinuation Rates and Patterns
Most patients with overweight or obesity discontinued GLP-1 RA therapy within one year, according to cohort study data. Patients without type 2 diabetes had higher discontinuation rates and lower reinitiation rates compared to those with diabetes.
These patterns reveal a troubling reality: inequities in access and adherence to effective treatments have the potential to exacerbate disparities in obesity management.
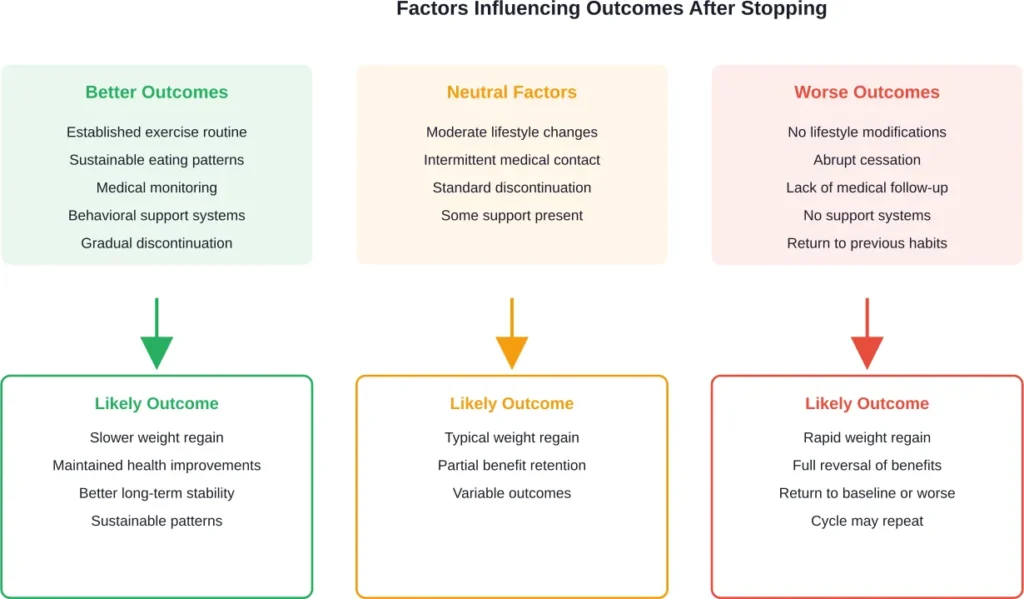
How to Safely Stop Taking Ozempic
Abrupt discontinuation isn’t necessarily dangerous for most people, but strategic approaches may help maintain some benefits.
Consult Healthcare Providers First
Never stop without medical guidance. Doctors can adjust dosages gradually, monitor blood sugar closely, and prepare alternative treatment plans.
For diabetes patients, transitioning to other medications prevents dangerous glucose spikes.
Strengthen Lifestyle Foundations
The medication works best alongside behavioral changes. Strengthening these habits before discontinuation may slow—though rarely prevent—weight regain.
Focus on sustainable nutrition patterns rather than restrictive diets. Regular physical activity, particularly resistance training, helps preserve muscle mass during and after treatment.
Consider Maintenance Strategies
Research on paths for discontinuation while maintaining weight loss remains limited. Some strategies under investigation include:
- Gradual dose tapering rather than abrupt stopping
- Intermittent dosing schedules
- Transitioning to other weight management medications
- Intensive behavioral intervention programs
None of these approaches have proven consistently effective at preventing weight regain, but they may offer modest benefits for some individuals.
The Reality of Chronic Treatment
Here’s the thing though—obesity behaves like a chronic condition.
Research findings confirm this reality. The chronicity of obesity suggests ongoing treatment is required to maintain weight reduction, similar to how hypertension or diabetes need continuous management.
This doesn’t mean permanent dependence on any single medication. But it does mean weight maintenance typically requires sustained intervention, whether pharmaceutical, behavioral, surgical, or combined.
Mental Health Considerations
The FDA has been monitoring reports of mental health effects with GLP-1 RAs. While preliminary evaluation doesn’t suggest a causal link between these medications and suicidal thoughts, patients should remain vigilant.
Tell healthcare professionals about any new or worsening depression, suicidal thoughts, or unusual changes in mood or behavior. Call or text 988 or visit https://988lifeline.org/ for free support 24/7.
The psychological impact of weight regain after stopping also deserves attention. Many patients experience frustration, disappointment, or feelings of failure when weight returns despite their best efforts.
Frequently Asked Questions
Weight regain typically begins within weeks of discontinuation. Research shows participants regain approximately two-thirds of their prior weight loss within one year after stopping semaglutide. The rate varies individually based on lifestyle factors, metabolic health, and genetic predisposition.
Most people can stop Ozempic suddenly without dangerous withdrawal effects, but gradual tapering under medical supervision may help minimize blood sugar fluctuations for diabetes patients. Consult healthcare providers before making any changes to medication schedules. They can monitor transitions and adjust other treatments as needed.
Blood sugar levels typically begin rising within days to weeks after discontinuation in patients with type 2 diabetes. The timeline depends on individual metabolic factors and whether alternative diabetes medications are implemented. Regular monitoring becomes essential during this transition period.
Research indicates that maintaining weight loss after stopping semaglutide is challenging for most patients. Studies show participants regain two-thirds of lost weight within one year of discontinuation. Individual results vary, but complete maintenance without continued intervention remains uncommon based on current evidence.
Long-term safety data continues accumulating as these medications see wider use. Common side effects include gastrointestinal complaints affecting approximately 62% of users. More serious but rare effects like gastroparesis have been reported. The FDA continues evaluating safety signals including mental health effects, though no causal links have been established for most concerns.
Patients can typically restart Ozempic after stopping, though this should be done under medical supervision. Research shows patients without type 2 diabetes have lower reinitiation rates compared to those with diabetes, possibly due to cost and insurance coverage barriers. Restarting usually follows the original dose escalation schedule.
Ozempic doesn’t cause traditional withdrawal symptoms like substances of abuse. However, patients commonly experience physiological changes including increased appetite, energy fluctuations, and metabolic shifts as the medication’s effects wear off. These represent a return to pre-treatment physiology rather than true withdrawal.
Moving Forward After Discontinuation
Stopping Ozempic doesn’t mean abandoning health goals entirely.
The experience of weight loss—even if temporary—can provide valuable insights about eating patterns, hunger signals, and sustainable habits. Some patients use this knowledge to build lasting behavioral changes.
Medical supervision remains important. Regular monitoring helps catch concerning changes in blood sugar, blood pressure, or other health markers. Healthcare providers can recommend alternative treatments or interventions to maintain some benefits.
Real talk: obesity treatment remains imperfect. Current evidence suggests most patients need ongoing pharmaceutical intervention to maintain significant weight loss. That’s not personal failure—it’s biological reality.
Understanding what happens after stopping Ozempic helps set realistic expectations. The two-thirds weight regain statistic isn’t meant to discourage, but to inform treatment planning. Patients and providers can make better decisions about long-term strategies when armed with honest data about discontinuation outcomes.
For those considering stopping or who’ve already discontinued treatment, remember that healthcare is a journey, not a destination. Whether continuing medication, trying alternative approaches, or focusing on non-scale health markers, informed choices lead to better outcomes than unrealistic expectations.
